• evaluating nominations by stockholders of candidates for election to our Board of Directors;
• considering and assessing the independence of members of our Board of Directors;
• developing a set of corporate governance policies and principles, periodically reviewing and assessing these policies and principles and their application and recommending to our Board of Directors any changes to such policies and principles;
• considering questions of possible conflicts of interest of directors as such questions arise; and
• reviewing and evaluating on an annual basis the performance of the Nominating and Corporate Governance Committee and the Nominating and Corporate Governance Committee charter.
In addition, in December 2021, we updated the charter for the Nominating and Corporate Governance Committee to provide for oversight of environmental, social and governance ("ESG") matters.
The current members of the Nominating and Corporate Governance Committee are Ms. Messemer, Mr. Sisitsky and Dr. Witte. Our Board has determined that each of the members of this committee is independent (as independence is currently defined in Rule 5605(a)(2) of the Nasdaq listing standards). Dr. Witte serves as the chair of our Nominating and Corporate Governance Committee.
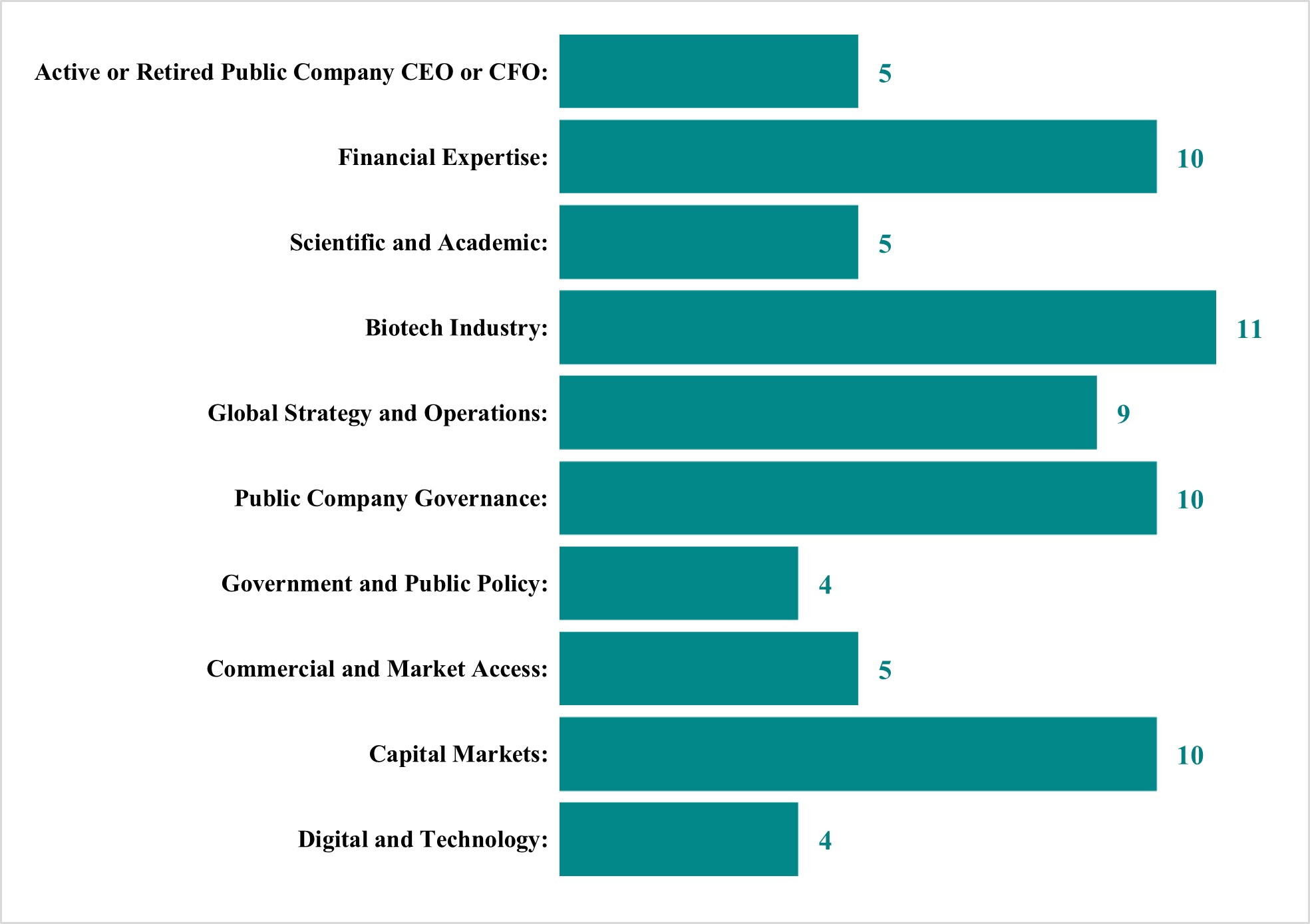
The Nominating and Corporate Governance Committee believes that the candidates for director, both individually and collectively, have the integrity, experience, judgment, commitment (including having sufficient time to devote to us and level of participation), skills, diversity and expertise appropriate for us. In assessing the directors, both individually and collectively, the Nominating and Corporate Governance Committee considers our current needs, and the needs of the Board of Directors, to maintain a balance of knowledge, experience, capability, race, gender, geography, thought, viewpoints, backgrounds, skills, and expertise. However, the Nominating and Corporate Governance Committee retains the right to modify these qualifications from time to time.
The Nominating and Corporate Governance Committee uses its network of contacts to compile a list of potential candidates, but may also engage, if it deems appropriate, a professional search firm. Candidates for director nominees are reviewed in the context of the current composition of the Board of Directors, our operating requirements and the long-term interests of stockholders. In conducting this assessment, the Nominating and Corporate Governance Committee typically considers diversity (including with respect to race, gender, geography, thought, viewpoints, and backgrounds), age, skills and such other factors as it deems appropriate given our current needs and the needs of the Board of Directors, to maintain a balance of knowledge, experience and capability. In the case of incumbent directors whose terms of office are set to expire, the Nominating and Corporate Governance Committee reviews these directors’ overall service to us during their terms, including the number of meetings attended, level of participation, quality of performance and any other relationships and transactions that might impair the directors’ independence. In the case of new director candidates, the Nominating and Corporate Governance Committee also determines whether the nominee is independent for Nasdaq purposes, which determination is based upon applicable Nasdaq listing standards, applicable SEC rules and regulations and the advice
of counsel, if necessary.
The Nominating and Corporate Governance Committee then uses its network of contacts to compile a list of potential candidates, but may also engage, if it deems appropriate, a professionalTo the extent any search
firm. Any search firm
is retained to assist the Nominating and Corporate Governance Committee in seeking candidates for the Board,
the search firm will be instructed to seek to include diverse candidates in terms of race, gender, geography, thought, viewpoints, backgrounds, skills, experience, and expertise from, among other areas, professional and academic areas relevant to the Company’s area of focus. In addition, the Nominating and Corporate Governance Committee conducts any appropriate and necessary inquiries into the backgrounds and qualifications of possible candidates after considering the function and needs of the Board of Directors. The Nominating and Corporate Governance Committee meets to discuss and consider the candidates’ qualifications and then selects a nominee for recommendation to the Board of Directors by majority vote.
The Nominating and Corporate Governance Committee will consider director candidates recommended by stockholders. The Nominating and Corporate Governance Committee does not intend to alter the manner in which it evaluates candidates, including the minimum criteria set forth above, based on whether or not the candidate was recommended by a stockholder. Stockholders who wish to recommend individuals for consideration by the Nominating and Corporate Governance Committee to become nominees for election to the Board of Directors may do so by delivering a written recommendation to the Nominating and Corporate
16
Governance Committee at the following address: 210 East Grand Avenue, South San Francisco, California 94080, Attn: Secretary, no later than the 90th day and no earlier than the 120th day prior to the one year anniversary of the preceding year’s Annual Meeting. Submissions must include, among other things, (1) the name and address of the stockholder on whose behalf the submission is made; (2) number of our shares that are owned beneficially by such stockholder as of the date of the submission; (3) the full name of the proposed candidate; (4) description of the proposed candidate’s business experience for at least the previous five years; (5) complete biographical information for the proposed candidate; (6) a description of the proposed candidate’s qualifications as a director and (7) any other information required by our Amended and Restated Bylaws. We may require any proposed nominee to furnish such other information as we may reasonably require to determine the eligibility of such proposed nominee to serve as our independent director or that could be material to a reasonable stockholder’s understanding of the independence, or lack thereof, of such proposed nominee.
The Nominating and Corporate Governance Committee charter can be found on our website at www.allogene.com.
International and Business Development Oversight Committee
Our International and Business Development Oversight Committee consists of Dr. Humer, as chair, and Mr. DeYoung, Mr. Kazam and Dr. Sato. The purpose of the International and Business Development Oversight Committee is to assist our Board of Directors in fulfilling our Board of Directors' responsibilities to (1) oversee our international and business development strategic planning, and (2) review and evaluate any of our specific strategic initiatives, including the risks and opportunities relating to such initiatives. The International and Business Development Oversight Committee charter can be found on our website at www.allogene.com.
Stockholder Communications With The Board Of Directors
The Company’s Board has adopted a formal process by which stockholders may communicate with the Board or any of its directors. Stockholders who wish to communicate with the Board may do so by sending written communications addressed to: Attn: Secretary, 210 East Grand Avenue, South San Francisco, California 94080. These communications will be reviewed by the Secretary, who will determine whether the communication is appropriate for presentation to the Board or the relevant director. The purpose of this screening is to allow the Board to avoid having to consider irrelevant or inappropriate communications (such as advertisements, solicitations and hostile communications).
The Company has adopted the Code of Business Conduct and Ethics that applies to all officers, directors and employees. The Code of Business Conduct and Ethics is available on the Company’s website at www.allogene.com. If the Company makes any substantive amendments to the Code of Business Conduct and Ethics or grants any waiver from a provision of the Code to any executive officer or director, the Company will promptly disclose the nature of the amendment or waiver on its website.
17
Advisory Vote on Executive Compensation
At our 2020 Annual Meeting of Stockholders, the stockholders indicated their preference that we solicit a non-binding advisory vote on the compensation of our named executive officers, commonly referred to as a “say-on-pay vote,” every year. The Board has adopted a policy that is consistent with that preference. In accordance with that policy, this year, we are again asking the stockholders to approve, on an advisory basis, the compensation of our named executive officers as disclosed in this proxy statement in accordance with SEC rules.
This vote is not intended to address any specific item of compensation, but rather the overall compensation of the Company’s named executive officers and the philosophy, policies and practices described in this proxy statement. The compensation of the Company’s named executive officers subject to the vote is disclosed in the Compensation Discussion and Analysis, the compensation tables, and the related narrative disclosure contained in this proxy statement. As discussed in those disclosures, we believe that our compensation policies and decisions are performance driven and foster a performance-oriented culture, are strongly aligned with our stockholders’ interests and are consistent with current market practices. Compensation of the Company’s named executive officers is designed to enable the Company to attract and retain talented and experienced executives to lead the Company successfully in a competitive environment.
In light of our performance in 2021, we believe that the compensation paid to our named executive officers was appropriate. In 2021, we advanced and reported interim data from our lead product candidates, ALLO-501, ALLO-501A and ALLO-715, initiated clinical trials of ALLO-605 and ALLO-316, commenced current good manufacturing practices ("GMP") manufacturing at our manufacturing facility, advanced our innovative research, expanded our board of directors and scientific advisory board, and progressed our joint venture in China, each as further described in the summary of our executive compensation disclosure contained in this proxy statement. In addition, in October 2021, we faced a clinical hold on all of our clinical trials. Through exceptional technical leadership and diligence, we resolved the clinical hold in January 2022. The Board and Compensation Committee refrained from rewarding any additional compensation, including any 2021 annual incentive bonus, until the clinical hold was resolved.
Accordingly, the Board is asking the stockholders to indicate their support for the compensation of the Company’s named executive officers as described in this proxy statement by casting a non-binding advisory vote “FOR” the following resolution:
“RESOLVED, that the compensation paid to the Company’s named executive officers, as disclosed pursuant to Item 402 of Regulation S-K, including the Compensation Discussion and Analysis, compensation tables and narrative discussion is hereby APPROVED.”
Because the vote is advisory, it is not binding on the Board or the Company. Nevertheless, the views expressed by the stockholders, whether through this vote or otherwise, are important to management and the Board and, accordingly, the Board and the Compensation Committee intend to consider the results of this vote in making determinations in the future regarding executive compensation arrangements.
Advisory approval of this proposal requires the vote of the holders of a majority of the shares present at the Annual Meeting or represented by proxy and entitled to vote on the matter at the Annual Meeting. Unless the Board decides to modify its policy regarding the frequency of soliciting advisory votes on the compensation of the Company’s named executive officers, the next scheduled say-on-pay vote will be at the 2023 Annual Meeting of Stockholders.
The Board of Directors Recommends
A Vote “For” Proposal 2.
Proposal 3
Approval of the Increase in Number of Authorized Shares of Common Stock
General
The Board has approved an amendment (the “Authorized Shares Amendment”) to the Company’s Amended and Restated Certificate of Incorporation (the “Certificate of Incorporation”) to increase the number of authorized shares of common stock from 200,000,000 to 400,000,000. The Authorized Shares Amendment will not change the number of authorized shares of preferred stock, which currently consists of 10,000,000 shares of preferred stock.
The additional shares of common stock authorized for issuance by the Authorized Shares Amendment would be a part of the existing class of common stock and, if and when issued, would have the same rights and privileges as the common stock presently issued and outstanding. The full text of the proposed Authorized Shares Amendment is attached to this Proxy Statement as Appendix A. However, the text of the Authorized Shares Amendment is subject to revision to include such changes as may be required by the Secretary of State of the State of Delaware and as deemed necessary and advisable to effect the Authorized Shares Amendment.
Provided the stockholders approve the Authorized Shares Amendment, the increased number of shares would be authorized for issuance, but would remain unissued until such time as the Board approves a specific issuance of such shares. Other than future issuances under the Company’s equity compensation plans and issuances of common stock that may be issued and sold under our Common Stock Sales Agreement with Cowen and Company, LLC ($167.3 million of common stock remains available for sale under such agreement as of December 31, 2021), the Company currently has no plans or arrangements to issue the additional authorized shares of common stock that will result in the event that the Company’s stockholders approve, and the Company implements, the Authorized Shares Amendment.
Adoption of the Authorized Shares Amendment would not affect the rights of the holders of currently outstanding common stock, except for effects incidental to increasing the number of shares of our common stock outstanding, such as dilution of the earnings per share and voting rights of current holders of common stock, to the extent that any additional shares of common stock are ultimately issued out of the increase in authorized shares proposed in the Authorized Shares Amendment.
If the proposed Authorized Shares Amendment is approved by the requisite vote of the stockholders, it will become effective upon the filing of a Certificate of Amendment with the Secretary of State of the State of Delaware. The Board reserves its right to elect not to proceed with and abandon the Authorized Shares Amendment if it determines, in its sole discretion at any time, that this proposal is no longer in the best interests of our stockholders.
If we fail to obtain stockholder approval of this proposal at the Annual Meeting, we may continue to seek to obtain stockholder approval at each subsequent annual meeting of stockholders and/or special meetings of stockholders until such approval has been obtained and we will incur the costs associated therewith.
Background
In addition to the 143,569,902 shares of common stock outstanding on March 31, 2022, we have also reserved, as of March 31, 2022, 22,567,690 shares for issuance upon the exercise or vesting of outstanding stock awards and 19,889,713 shares for issuance pursuant to the Company’s equity incentive and employee stock purchase plans, meaning that we presently have 13,972,695 authorized shares available for issuance, which is insufficient to meet our needs in connection with future financings or strategic transactions and properly incentivizing our key personnel.
Purposes and Effects of the Authorized Shares Amendment
The Board is recommending the proposed increase in the authorized number of shares of common stock to provide the Company with appropriate flexibility to issue additional shares in the future on a timely basis if such need arises in connection with potential financings, business combinations or other corporate purposes. Approval of the Authorized Shares Amendment could enable the Company to take advantage of market conditions, the availability of more favorable financing, and opportunities for business combinations and other strategic transactions, without the potential delay and expense associated with convening a special stockholders’ meeting.
Our success also depends in part on our continued ability to attract, retain and motivate highly qualified management and key personnel. If this proposal is not approved by our stockholders, the lack of unissued and unreserved authorized shares of common stock to provide future equity incentive opportunities could adversely impact our ability to achieve these goals. In short, if our
stockholders do not approve this proposal, we may not be able to access the capital markets, complete corporate collaborations or partnerships, attract, retain and motivate employees, and pursue other business opportunities integral to our growth and success.
The proposed increase in the number of authorized shares of common stock will not, by itself, have an immediate dilutive effect on our current stockholders. However, if this proposal is approved, unless otherwise required by applicable law or stock exchange rules, the Board will be able to issue the additional shares of common stock from time to time in its discretion without further action or authorization by the stockholders. The newly authorized shares of common stock would be issuable for any proper corporate purpose, including capital raising transactions of equity or convertible debt securities, the establishment of collaborations or other strategic agreements, stock splits, stock dividends, issuance under current or future equity incentive plans, future acquisitions, investment opportunities, or for other corporate purposes. The future issuance of additional shares of common stock or securities convertible into our common stock may occur at times or under circumstances that could result in a dilutive effect on the earnings per share, book value per share, voting power and percentage interest of the present holders of our common stock, some of whom have preemptive rights to subscribe for additional shares that we may issue.
Potential Anti-Takeover Effect
An increase in the number of authorized but unissued shares of common stock relative to the number of outstanding shares of common stock may also, under certain circumstances, be construed as having an anti-takeover effect. Although not designed or intended for such purposes, the effect of the Authorized Shares Amendment might be to render more difficult or to discourage a merger, tender offer, proxy contest or change in control of us and the removal of management, which stockholders might otherwise deem favorable. For example, the authority of the Board to issue common stock might be used to create voting impediments or to frustrate an attempt by another person or entity to effect a takeover or otherwise gain control of us because the issuance of additional common stock would dilute the voting power of the common stock then outstanding. Our common stock could also be issued to purchasers who would support the Board in opposing a takeover bid which our Board determines not to be in our best interests and those of our stockholders. In addition to the Authorized Shares Amendment, the Certificate of Incorporation and Amended and Restated Bylaws also include other provisions that may have an anti-takeover effect. These provisions, among other things, permit the Board to issue preferred stock with rights senior to those of the common stock without any further vote or action by the stockholders, provide that special meetings of stockholders may only be called by the Board and some of our officers, and do not provide for cumulative voting rights, which could make it more difficult for stockholders to effect certain corporation actions and may delay or discourage a change in control. The Board is not presently aware of any attempt, or contemplated attempt, to acquire control of the Company and the Authorized Shares Amendment is not part of any plan by the Board to recommend or implement a series of anti-takeover measures.
Vote Required
Approval of this proposal requires the affirmative vote of a majority of the shares of our common stock outstanding on the record date for the Annual Meeting. Abstentions will have the same effect as an “against” vote on this proposal. As noted above, we believe that this proposal will be considered a “routine” matter and, as a result, we do not expect there to be any broker non-votes on this proposal. If, however, a broker non-vote occurs (or if your shares are not affirmatively voted in favor of this proposal for any other reason), it will have the same effect as an “against” vote on this proposal.
The Board of Directors Recommends
A Vote “For” Proposal 3.
Proposal 4
Approval of the Stock Option Exchange Program
On April 13, 2022, our Board authorized a stock option exchange program (the “Option Exchange”) pursuant to which our eligible employees would be given the opportunity to exchange eligible stock options for new stock options with an exercise price equal to the fair market value of our common stock at the time of the exchange. An eligible stock option generally includes any employee stock option that has an exercise price equal to or greater than $18.00 per share and was granted on or prior to December 31, 2021 pursuant to our 2018 Amended and Restated Equity Incentive Plan (the “2018 Plan”).
As of March 31, 2022, we had outstanding stock options held by employees to purchase 6,654,366 shares of common stock with a weighted average exercise price of $19.36 per share (exclusive of the Excluded Employees as defined below). Of these employee stock options, there were 4,070,873 shares with an exercise price equal to or greater than $18.00 per share with a weighted-average exercise price of $26.78 that would be considered eligible stock options for purposes of the Option Exchange.
The Board believes that the Option Exchange is in the best interests of stockholders and the Company, as new stock options granted under the program will provide added incentive to motivate and retain our talented employees.
Rationale for Option Exchange
We evaluated several alternatives for remaining competitive within our industry and with our employees, including increasing cash compensation and/or granting additional equity awards. While these components are part of our overall compensation packages, we do not believe that relying exclusively on such approaches is an ideal use of our resources. For example, increasing cash compensation would reduce the cash resources we devote to research and development, and granting additional stock awards would cause dilution to our current stockholders and greater expense. Accordingly, we determined that the Option Exchange was the most attractive alternative for stockholders for the reasons set forth below.
Employee Retention, Motivation and Performance
We have designed the Option Exchange to restore equity value, increase retention and motivation in a competitive labor market, provide non-cash compensation incentives and align our employee and stockholder interests for long-term value creation. Underwater stock option awards are of limited benefit in motivating and retaining our employees. Through the Option Exchange, we believe that we will be able to enhance long-term stockholder value by increasing our ability to retain experienced and talented employees and by aligning the interests of these individuals more fully with the interests of our stockholders.
Because 61% of our eligible employee stock options are underwater (and for a large number of employees, significantly so), as of March 31, 2022, we may face a considerable challenge in retaining our employees and there is a possibility that our competitors may be able to offer equity incentives that are more attractive, which in some cases, could make the terms of employment at a new employer more attractive than we can offer to our existing employees. Additionally, the underwater options have less perceived value and are unlikely to incentivize employees to work harder and build up value in the Company. The Option Exchange is designed to address these concerns, improve morale among our employees generally and reinvigorate a culture where equity compensation is a key component of our overall compensation package.
As discuss in more detail below, none of the new stock options issued under the Option Exchange program will be vested on the date of grant. Stock options issued in the Option Exchange will vest in equal annual amounts over a three-year period from the grant date of such new stock options. The stock options eligible to be exchanged are generally subject to a four-year vesting schedule, in which twenty-five percent of shares vest after one year, and the remaining shares vest in equal monthly installments. Our Compensation Committee believes that implementing a new extended vesting schedule is appropriate because it encourages retention of employees over the next three years, during a highly critical period for the Company.
Impact on Compensation Expense
The “fair value” of the stock options eligible for exchange was based on the then fair market value of our common stock on the applicable grant date. Under applicable accounting rules, we will recognize a total of approximately $73.9 million in non-cash compensation expense related to these underwater stock options, $36.3 million of which has already been expensed as of December 31, 2021 and $37.6 million of which we will continue to be obligated to expense, even if these stock options are never exercised because they remain underwater. Assuming that the exercise price of the replacement options will be equal to $9.11 per share (which was the fair market value of our stock on March 31, 2022), the replacement took effect on March 31, 2022 and all eligible employees participate, and that the inputs for the Black Scholes option pricing model used are held constant, the replacement options will result in an additional non-cash compensation expense of approximately $8.7 million. By replacing current stock options that have little or no
retention or incentive value with new stock options that will provide both retention and incentive value while being mindful of the additional compensation expense, we believe that we will be making efficient use of our resources.
Alternatives Considered
Our Compensation Committee considered alternatives to the Option Exchange to provide meaningful performance and retention incentive to our employees, including providing new equity to employees, repricing the options with no change in vesting, exchanging underwater options for full value shares, or increasing cash compensation. The Compensation Committee determined that the Option Exchange provides better performance and retention incentives with less cost to the Company or dilution to stockholders than the other alternatives.
Structure of the Option Exchange
The Board authorized the Option Exchange on April 13, 2022, subject to stockholder approval. It is currently anticipated that the Option Exchange will commence as soon as reasonably practicable following approval of this Proposal 4 by our stockholders, or the Commencement Date. At the start of the Option Exchange, employees holding eligible stock options will receive a written exchange offer that will set forth the precise terms of the Option Exchange. The written offer will be governed by the tender offer rules of the SEC. At or before the Commencement Date, we will file the offer to exchange and other related documents with the SEC as part of a tender offer statement on Schedule TO. Eligible optionholders will be given at least 20 business days to elect to participate in the Option Exchange. Eligible optionholders must choose to participate in the program for all of their eligible options or none, and may not choose to exchange portions of eligible option grants or some eligible option grants and not others. Set forth below is a description of the key features of the Option Exchange.
Eligible Participants
The Option Exchange will be available to employees, excluding our executive officers and Chief Communications Officer (collectively, the "Excluded Employees"), who on the Commencement Date are employed by us and hold outstanding eligible stock options. As of March 31, 2022, eligible stock options were held by approximately 71% of employees (exclusive of the Excluded Employees). Participants in the Option Exchange must continue to be employed by and provide services to us on the date the surrendered options are cancelled and replacement stock options are granted. Any employee holding eligible stock options who elects to participate in the Option Exchange but whose service with us terminates for any reason before the date the new stock options are granted, including a termination due to voluntary resignation, retirement, involuntary termination, layoff, death or disability, would retain his or her eligible options subject to their existing terms and will not be eligible to receive new stock options in the Option Exchange.
Eligible Stock Options
An eligible stock option generally includes any eligible employee stock option that has an exercise price equal to or greater than $18.00 per share and was granted on or prior to December 31, 2021. The $18.00 price threshold represents an approximately 200% premium to the volume weighted average stock price for 30 trading days through April 13, 2022. As of March 31, 2022, we had outstanding eligible stock options to purchase 4,070,873 shares of common stock under the 2018 Plan at a weighted-average exercise price of $26.78 per share and with a weighted-average remaining life of 7.9 years. These eligible stock options represent approximately 2.8% of the issued and outstanding shares of our common stock as of March 31, 2022.
Exchange Ratio
The Option Exchange is a one-for-one exchange. Thus, each eligible option will be replaced by a new option covering the same number of shares, but with a new exercise price, term, and vesting schedule. The overall number of stock options will stay the same.
Vesting Schedules for New Options
New stock option awards will not be vested on the date of grant. Eligible stock options may be exchanged for new stock options with a new three-year vesting schedule, in each case vesting in equal annual installments over the vesting term. These new vesting schedules support the nature of stock options as an incentive vehicle and provide us with valuable additional years of employee retention during a highly critical time for the Company.
Term for New Options
The new stock options will expire seven (7) years following the date the new options are granted.
Intended Implementation of the Option Exchange As Soon As Practicable Following Stockholder Approval
We expect that the Option Exchange will begin as soon as reasonably practicable, but no later than four (4) months following stockholder approval, if received. Our Board in its discretion reserves the right to amend, postpone or, under certain circumstances, cancel the Option Exchange once it has commenced, but the Option Exchange will not be materially amended in a manner more beneficial to eligible participants without first seeking additional stockholder approval.
Impact of Option Exchange on Surrendered Options
Under the terms of the Option Exchange, there will be no shares returned to the share reserve of the 2018 Plan because it will be a one-for-one exchange.
Option Exchange Process
Additional information about how we expect to conduct the Option Exchange, if approved by stockholders, is set forth below. While the terms of the Option Exchange are expected to conform to the material terms described above in this Proposal 4, we may find it necessary or appropriate to change the terms of the Option Exchange to take into account our administrative needs, accounting rules, company policy decisions or to comply with any comments we receive from the SEC. We may decide not to implement the Option Exchange even if stockholder approval of the Option Exchange is obtained, or we may delay, amend or terminate the Option Exchange once it is in progress. The final terms of the Option Exchange will be described in the exchange offer documents that will be filed with the SEC and available at www.sec.gov.
Overview of the Option Exchange Process
Upon commencement of the Option Exchange, eligible participants holding eligible stock option awards will receive a written offer setting forth the terms of the Option Exchange and may voluntarily elect to participate. All eligible employees who are employed by us on the Commencement Date, are still employed by us on the grant date of the new stock options, and hold eligible stock option awards may participate in the Option Exchange. Eligible participants will be given at least 20 business days to elect to surrender eligible stock options in exchange for the same number of new stock options. Upon completion of the Option Exchange, surrendered stock options will be cancelled and new stock options will be granted. Cancelled options will then be available for future grant under the 2018 Plan.
The 2018 Plan will govern all terms or conditions of new stock options not specifically addressed by the Option Exchange described in this proxy statement. Additionally, it is anticipated that new options will be incentive stock options (that is, they will qualify for the tax-favored treatment) to the extent allowable under Section 422 of the Internal Revenue Code and available for grant under our 2018 Plan.
Election to Participate
Eligible participants will receive a tender offer document and will be able to voluntarily elect to participate in the Option Exchange. If you are both a stockholder and employee holding stock options that are potentially subject to the Option Exchange, note that voting to approve the Option Exchange does not constitute an election to participate in the Option Exchange. The written exchange offer documents described above will be provided if and when the Option Exchange is commenced, and you can only elect to participate after that time.
Impact of Option Exchange on Number of Options Issued
The Compensation Committee established a 1-to-1 exchange ratio, that will result in the issuance of the same number of stock options through the Option Exchange.
Effect on Stockholders
Under the terms of the Option Exchange, the new stock options will have an exercise price commensurate with their fair market value at the time of the Option Exchange, making the stock options no longer underwater. As stated in the “Rationale for Option Exchange” Section, these new stock options should help keep our valuable employees at our Company and motivate them to create long-term value for our Company and stockholders.
Interests of Our Executive Officers and Non-Employee Directors in the Option Exchange
The Excluded Employees and our non-employee directors (including our Executive Chair) will not be permitted to participate in the Option Exchange.
Accounting Impact
The incremental compensation cost associated with the Option Exchange will be measured as the excess, if any, of the fair value of each award of new stock option granted to participants in the Option Exchange, measured as of the date the new stock options are granted, over the fair value of the stock options surrendered in exchange for the new stock options, measured immediately prior to the cancellation. This incremental compensation cost will be recognized ratably over the vesting period of the new stock options.
Material U.S. Federal Income Tax Consequences of the Option Exchange
The exchange of stock options pursuant to the Option Exchange should be treated as a non-taxable exchange because the new stock options will have an exercise price equal to or greater than the fair market value of our common stock on the grant date. Neither the Company, nor participants in the Option Exchange, should recognize any income for U.S. federal income tax purposes upon the grant of the new stock options. To the extent permissible and available for grant under our 2018 Plan, new stock options granted under the Option Exchange will be incentive stock options for U.S. federal income tax purposes. Tax effects may vary in other countries; a more detailed summary of tax considerations will be provided to all participants in the Option Exchange documents.
Financial Statements
Our consolidated financial statements and other information required by Item 13(a) of Schedule 14A under the Exchange Act are incorporated by reference from our Annual Report on Form 10-K for the fiscal year ended December 31, 2021, filed with the SEC on February 23, 2022.
Vote Required
Approval of the Option Exchange requires “For” votes from a majority of the shares present or represented by proxy at the Annual Meeting and entitled to vote on this proposal. Abstentions will have the same effect as an “Against” vote on this proposal. Broker non-votes will have no effect.
If you are both a stockholder and an employee holding eligible stock options, please note that voting to approve this program does not constitute an election to participate in the program.
Our Recommendation
The Board of Directors Recommends
A Vote “For” Proposal 4.
Proposal 5
Ratification of Selection of Independent Registered Public Accounting Firm
The Audit Committee of the Board of Directors has selected Ernst & Young LLP as the Company’s independent registered public accounting firm for the fiscal year ending December 31,
20192022 and has further directed that management submit the selection of its independent registered public accounting firm for ratification by the stockholders at the
annual meeting.Annual Meeting. Ernst & Young LLP has audited the Company’s financial statements since its inception in 2017. Representatives of Ernst & Young LLP are expected to be present at the
annual meeting.Annual Meeting. They will have an opportunity to make a statement if they so desire and will be available to respond to appropriate questions.
Neither the Company’s Bylaws nor other governing documents or law require stockholder ratification of the selection of Ernst & Young LLP as the Company’s independent registered public accounting firm. However, the Audit Committee of the Board is submitting the selection of Ernst & Young LLP to the stockholders for ratification as a matter of good corporate practice. If the stockholders fail to ratify the selection, the Audit Committee of the Board will reconsider whether or not to retain that firm. Even if the selection is ratified, the Audit Committee of the Board in its discretion may direct the appointment of a different independent registered public accounting firm at any time during the year if they determine that such a change would be in the best interests of the Company and its stockholders.
The affirmative vote of the holders of a majority of the shares present
in personat the Annual Meeting or represented by proxy and entitled to vote on the matter at the
annual meetingAnnual Meeting will be required to ratify the selection of
the accounting firm.
Abstentions will have the same effect as an “against” vote on this proposal. Broker non-votes, if any, will have no effect.
Principal Accountant Fees and Services
The following table represents aggregate fees billed to the Company for the
period from November 30, 2017 (inception) through December 31, 2017 and for the fiscal
yearyears ended December 31,
20182020 and December 31, 2021 by Ernst & Young LLP, the Company’s principal accountant.
| | | | | | | | |
| | | Fiscal Year
Ended
2018 | | | Period from
November 30, 2017
(Inception) to
December 31, 2017 | |
| | | (in thousands) | |
Fee Category | | | | | | | | |
Audit fees(1) | | $ | 1,518 | | | $ | 26 | |
Audit-related fees(2) | | | 75 | | | | — | |
Tax fees | | | — | | | | — | |
All other fees(3) | | | 2 | | | | — | |
| | | | | | | | |
Total fees | | $ | 1,595 | | | $ | 26 | |
| | | | | | | | |
(1) | Audit fees consist of fees for professional services provided primarily in connection with the annual audit of our financial statements, quarterly reviews and services associated with SEC registration statements and other documents issued in connection with the Company’s initial public offering including comfort letters and consents.
|
(2) | Audit-related fees consist of fees related to the adoption of a new lease accounting standard (codified in ASC 842).
|
(3) | All other fees consist of a subscription to Ernst & Young Atlas Online, a proprietary knowledge management and research system.
|
| | | | | | | | | | | |
| Fiscal Year Ended |
| 2021 | | 2020 |
| (in thousands) |
| Fee Category | | | |
| Audit fees(1) | $ | 1,580 | | $ | 1,331 |
| Audit-related fees | — | | — |
| Tax fees | — | | — |
| All other fees(2) | 2 | | 3 |
| Total fees | $ | 1,582 | | $ | 1,334 |
(1) Audit fees consist of fees for professional services provided primarily in connection with the annual audit of our financial statements, audit of our internal controls over financial reporting, quarterly reviews and services associated with SEC registration statements and other documents issued in connection with the Company’s at the market offerings, including comfort letters and consents.
(2) All other fees consist of a subscription to Ernst & Young Atlas Online, a proprietary knowledge management and research system.
All fees described above were
pre-approved by the Audit Committee.
18
Pre-Approval Policies and Procedures.
Pursuant to its charter, the Audit Committee must review and approve, in advance, the scope and plans for the audits and the audit fees and approve in advance (or, where permitted under the rules and regulations of the SEC, subsequently) all
non-audit services to be performed by the independent registered public accounting firm that are not otherwise prohibited by law and any associated fees. The Audit Committee may delegate to one or more members of the committee the authority to
pre-approve audit and permissible
non-audit services, as long as this
pre-approval is presented to the full committee at scheduled meetings.
The Board
Ofof Directors Recommends
A Vote “For” Proposal
2.19
5.
Proposal 6
Approval of an Adjournment of the Annual Meeting, if Necessary, to Solicit Additional Proxies
General
If the Annual Meeting is convened and a quorum is present, but there are not sufficient votes to approve Proposal 3, or if there are insufficient votes to constitute a quorum, our proxy holders may move to adjourn the Annual Meeting at that time in order to enable the Board to solicit additional proxies.
In this proposal, we are asking our stockholders to authorize the holder of any proxy solicited by the Board to vote in favor of adjourning the Annual Meeting to another time and place, if necessary or appropriate (as determined in good faith by the Board), to solicit additional proxies in the event there are not sufficient votes to approve Proposal 3. If our stockholders approve this proposal, we could adjourn the Annual Meeting and any adjourned or postponed session of the Annual Meeting and use the additional time to solicit additional proxies, including the solicitation of proxies from our stockholders that have previously voted. Among other things, approval of this proposal could mean that, even if we had received proxies representing a sufficient number of votes to defeat Proposal 3, we could adjourn the Annual Meeting without a vote on such proposal and seek to convince our stockholders to change their votes in favor of such proposal.
If it is necessary or appropriate (as determined in good faith by the Board) to adjourn the Annual Meeting, no notice of the adjourned meeting is required to be given to our stockholders, other than an announcement at the Annual Meeting of the time and place to which the Annual Meeting is adjourned, so long as the meeting is adjourned for 30 days or less and no new record date is fixed for the adjourned meeting. At the adjourned meeting, we may transact any business which might have been transacted at the original meeting.
Vote Required
Approval of this proposal requires the vote of the holders of a majority of the shares present at the Annual Meeting or represented by proxy and entitled to vote on the matter at the Annual Meeting. Abstentions will have the same effect as an “against” vote on this proposal. Broker non-votes, if any, will have no effect.
The Board of Directors Recommends
A Vote “For” Proposal 6.
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth certain information regarding the ownership of the Company’s common stock as of March
15, 201931, 2022 by: (i) each director; (ii) each of the Company’s named executive officers; (iii) all executive officers and directors of the Company as a group; and (iv) all those known by the Company to be beneficial owners of more than 5% of its common stock.
The table is based upon information supplied by officers, directors and principal stockholders, Schedules 13D and 13G filed with the SEC and other sources believed to be reliable by the Company. Unless otherwise indicated in the footnotes to this table and subject to community property laws where applicable, the Company believes that each of the stockholders named in this table has sole voting and investment power with respect to the shares indicated as beneficially owned. Applicable percentages are based on
121,527,320143,569,902 shares outstanding on March
15, 2019,31, 2022, adjusted as required by rules promulgated by the SEC. The number of shares of common stock used to calculate the percentage ownership of each listed beneficial owner includes the shares of common stock underlying options or convertible securities held by such beneficial owner that are exercisable or convertible within 60 days following March
15, 2019.31, 2022. Unless otherwise indicated, the address for each person or entity listed in the table is c/o Allogene Therapeutics, Inc., 210 East Grand Avenue, South San Francisco, California 94080.
| | | | | | | | |
Name of Beneficial Owner | | Number of
shares
Beneficially
Owned | | | Percentage
Beneficially
Owned | |
Greater than 5% Stockholders | | | | | | | | |
Pfizer Inc. (1) | | | 22,032,040 | | | | 18.1 | % |
Entities affiliated with TPG Group Holdings (SBS) Advisors, Inc.(2) | | | 23,135,061 | | | | 19.0 | % |
Gilead Sciences, Inc. (3) | | | 7,486,689 | | | | 6.2 | % |
Seaview Trust (4) | | | 8,514,287 | | | | 7.0 | % |
Directors and Named Executive Officers | | | | | | | | |
David Chang, M.D., Ph.D. (5) | | | 5,047,881 | | | | 4.1 | % |
Eric Schmidt, Ph.D. (6) | | | 1,549,730 | | | | 1.3 | % |
Alison Moore, Ph.D.(7) | | | 976,507 | | | | * | % |
Joshua Kazam (8) | | | 1,390,651 | | | | 1.1 | % |
Arie Belldegrun, M.D., FACS (9) | | | 12,109,522 | | | | 9.9 | % |
Franz Humer, Ph.D. (10) | | | 258,620 | | | | * | % |
Owen Witte, M.D. (11) | | | 221,182 | | | | * | % |
David Bonderman (12) | | | 23,135,061 | | | | 19.0 | % |
Deborah Messemer(13) | | | 216,535 | | | | * | % |
Todd Sisitsky | | | — | | | | — | % |
John DeYoung | | | — | | | | — | % |
All current executive officers and directors as a group (11 persons) (14) | | | 45,051,057 | | | | 36.5 | % |
| | | | | | | | | | | | | | |
| Name of Beneficial Owner | | Number of shares Beneficially Owned | | Percentage Beneficially Owned |
| Greater than 5% Stockholders | | | | |
| Pfizer Inc.(1) | | 22,032,040 | | 15.3% |
| TPG GP A, LLC(2) | | 18,716,306 | | 13.0% |
| Directors and Named Executive Officers | | | | |
| David Bonderman(3) | | 18,716,306 | | 13.0% |
| Arie Belldegrun, M.D.(4) | | 8,858,860 | | 6.2% |
| David Chang, M.D., Ph.D.(5) | | 8,427,632 | | 5.9% |
| Joshua Kazam(6) | | 242,387 | | * |
| Franz Humer, Ph.D.(7) | | 314,966 | | * |
| Deborah Messemer(8) | | 287,215 | | * |
| Owen Witte, M.D.(9) | | 275,222 | | * |
| Elizabeth Barrett(10) | | 63,385 | | * |
| Vicki Sato, Ph.D.(11) | | 34,692 | | * |
| Todd Sisitsky | | — | | | — |
| John DeYoung | | — | | | — |
| Eric Schmidt, Ph.D.(12) | | 1,846,218 | | 1.3% |
| Alison Moore, Ph.D.(13) | | 1,680,278 | | * |
| Rafael Amado, M.D.(14) | | 764,418 | | * |
| Veer Bhavnagri(15) | | 1,017,493 | | * |
| All current executive officers and directors as a group (15 persons)(16) | | 42,529,072 | | 29.8% |
* | Represents beneficial ownership of less than 1%.
| |
|
(1) | Consists of 22,032,040 shares of common stock held by Pfizer Inc. (“Pfizer”). The address of Pfizer is 235 E. 42nd Street, New York, NY 10017. This information is based on the Schedule 13D filed on October 22, 2018 with the SEC.
|
(2) | TPG Group Holdings (SBS) Advisors, Inc., a Delaware corporation (“Group Advisors”), is the sole member of TPG Group Holdings (SBS) Advisors, LLC, a Delaware limited liability company, which is the general partner of TPG Group Holdings (SBS), L.P., a Delaware limited partnership, which is the sole member of TPG Holdings IA, LLC, a Delaware limited liability company, which is the general partner of TPG Holdings I, L.P., a Delaware limited partnership, which is the sole member of each of (i) TPG GenPar VII Advisors, LLC, a Delaware limited liability company and (ii) The Rise Fund GenPar Advisors, LLC, a Delaware limited liability company. TPG GenPar VII Advisors, LLC is the general partner of TPG GenPar
|
20
* Represents beneficial ownership of less than 1%.
(1) Consists of 22,032,040 shares of common stock held by Pfizer Inc. (“Pfizer”). The address of Pfizer is 235 E. 42nd Street, New York, NY 10017. This information is based on a Form 4 filed by PF Equity Holdings 2 B.V., a wholly-owned subsidiary of Pfizer, on April 4, 2022 with the SEC.
(2) Consists of an aggregate of 18,716,306 shares of common stock. TPG GP A is the managing member of TPG Group Holdings (SBS) Advisors, LLC, a Delaware limited liability company, which is the general partner of TPG Group Holdings (SBS), L.P., a Delaware limited partnership, which holds 100% of the shares of Class B common stock (which represents a majority of the combined voting power of the common stock) of TPG Inc., a Delaware corporation (“TPG”), which is the controlling shareholder of TPG GP Co, Inc., a Delaware corporation, which is the managing member of TPG Holdings I-A, LLC, a Delaware limited liability company, which is the general partner of TPG Operating Group I, L.P., a Delaware limited partnership, which is the sole member of each of (i) TPG GenPar VII Advisors, LLC, a Delaware limited liability company and (ii) The Rise Fund GenPar Advisors, LLC, a Delaware limited liability company. TPG GenPar VII Advisors, LLC is the general partner of TPG GenPar VII, L.P., a Delaware limited partnership, which is the general partner of TPG Carthage Holdings, L.P., a Delaware limited partnership, which directly holds 12,477,536 shares of Common Stock. The Rise Fund GenPar Advisors, LLC is the general partner of The Rise Fund GenPar, L.P., a Delaware limited partnership, which it the general partner of The
| VII, L.P., a Delaware limited partnership, which is the general partner of TPG Carthage Holdings, L.P., a Delaware limited partnership, which directly holds 15,432,372 shares of common stock. The Rise Fund GenPar Advisors, LLC is the general partner of The Rise Fund GenPar, L.P., a Delaware limited partnership, which it the general partner of The Rise Fund Carthage, L.P., a Delaware limited partnership (together with TPG Carthage Holdings, L.P., the “TPG Funds”), which directly holds 7,711,689 shares of common stock. Because of Group Advisors’ relationship with the TPG Funds, Group Advisors may be deemed to beneficially own the shares of common stock held by the TPG Funds. David Bonderman and James G. Coulter are the sole shareholders of Group Advisors. Because of the relationship of Messrs. Bonderman and Coulter to Group Advisors, each of Messrs. Bonderman and Coulter may be deemed to beneficially own the shares of common stock held by the TPG Funds. Messrs. Bonderman and Coulter disclaim beneficial ownership of the shares of common stock held by the TPG Funds except to the extent of their pecuniary interest therein. The address of each of TPG Carthage, Rise Carthage and Group Advisors is c/o TPG Global, LLC, 301 Commerce Street, Suite 3300, Fort Worth, Texas 76102. This information is based on the Schedule 13D filed on October 25, 2018 with the SEC. |
(3) | Consists of 7,486,689 shares of common stock held by Gilead Sciences, Inc. (“Gilead”). The address of Gilead is 333 Lakeside Drive, Foster City, CA 94404. This information is based on the Schedule 13G filed on February 13, 2019 with the SEC.
|
(4) | Consists of 8,514,287 shares of common stock. Hanna Ackerman is trustee of the Seaview Trust and may therefore be deemed to be the beneficial owner of the common stock held by the Seaview Trust. Arie Belldegrun, M.D. FACS, is an economic beneficiary of the Seaview Trust, but he does not have voting or investment control over the common stock held by the Seaview Trust. The address of the Seaview Trust is 811 Strada Vecchia Road, Los Angeles, CA 90077. This information is based on the Schedule 13G filed on February 13, 2019 with the SEC.
|
(5) | Consists of (i) 1,674 shares of common stock held by David Chang, M.D., Ph.D., (ii) 1,101,838 shares of common stock held by the Chang 2006 Family Trust (“Chang Trust”), (iii) 848,022 shares of common stock held by the Julia Chang 2018 Irrevocable Trust (the “Julia Trust”), (iv) 848,022 shares of common stock held by the Robert Chang 2018 Irrevocable Trust (the “Robert Trust”), (v) 1,955,625 shares of common stock acquired by Dr. Chang upon the exercise of stock options, 1,425,977 of which will be subject to our right of repurchase as of 60 days of March 15, 2019, and (vi) 292,700 shares of common stock issuable upon exercise of options, all of which will be unvested but exercisable within 60 days of March 15, 2019. Dr. Chang isco-trustee
of the Chang Trust and trustee of the Julia Trust and Robert Trust.
|
(6) | Consists of (i) 1,480 shares of common stock held by Eric Schmidt, Ph.D., (ii) 1,464,750 shares of common stock held by the Eric Schmidt 2017 Family Irrevocable Trust (“Schmidt Trust”), which were acquired by Dr. Schmidt upon the exercise of stock options held by the Dr. Schmidt, all of which will be subject to our right of repurchase as of 60 days of March 15, 2019, and (iii) 83,500 shares of common stock issuable upon exercise of options, all of which will be unvested but exercisable within 60 days of March 15, 2019. Dr. Schmidt’s spouse is trustee of the Schmidt Trust.
|
(7) | Consists of (i) 507 shares of common stock held by Alison Moore, Ph.D., and (ii) 976,000 shares of common stock issuable upon exercise of options, all of which will be unvested but exercisable within 60 days of March 15, 2019 held by Dr. Moore.
|
(8) | Consists of 1,390,651 shares of common stock held by Joshua Kazam.
|
(9) | Consists of (i) 488,250 shares of common stock beneficially owned by Bellco Capital, LLC, of which Arie Belldegrun, M.D. FACS, is a manager, (ii) 3,710,006 shares of common stock beneficially owned by Arie and Rebecka Belldegrun, as Trustees of the Belldegrun Family Trust (“Belldegrun Trust”), (iii) 5,989,352 shares of common stock beneficially owned by VVAG Special Fund LLC (“VVAG”), of which VVAG LLC is the manager, of which Dr. Belldegrun serves as a senior managing director, (iv) 1,691,781 shares of common stock beneficially owned by Vida Ventures LLC (“Vida”), of which VV Manager LLC is the manager, of which Dr. Belldegrun is a Senior Managing Director, (v) 35,000 shares of common stock held by Dr. Belldegrun, and (vi) 195,133 shares of common stock issuable upon exercise of options, all of which will be unvested but exercisable within 60 days of March 15, 2019 held by Dr. Belldegrun. Dr. Belldegrun disclaims beneficial ownership of the shares held by VVAG and Vida, except to the extent of any pecuniary interest therein. The address of Dr. Belldegrun and the Belldegrun
|
21
Rise Fund Carthage, L.P., a Delaware limited partnership (together with TPG Carthage Holdings, L.P., the “TPG Funds”), which directly holds 6,238,770 shares of Common Stock. Because of TPG GP A’s relationship with the TPG Funds, TPG GP A may be deemed to beneficially own the shares of Common Stock held by the TPG Funds. TPG GP A is owned by entities owned by Messrs. Bonderman, Coulter and Winkelried. Because of the relationship of Messrs. Bonderman, Coulter and Winkelried to TPG GP A, each of Messrs. Bonderman, Coulter and Winkelried may be deemed to beneficially own the shares of Common Stock held by the TPG Funds. Messrs. Bonderman, Coulter and Winkelried disclaim beneficial ownership of the shares of Common Stock held by the TPG Funds except to the extent of their pecuniary interest therein. The address of each of the TPG Funds is c/o TPG Global, LLC, 301 Commerce Street, Suite 3300, Fort Worth, Texas 76102. This information is based on the Schedule 13D/A filed on January 18, 2022 with the SEC.
(3) Consists of the shares described in note (2) above.
(4) Consists of (i) 4,710,120 shares of common stock beneficially owned by Rebecka Belldegrun, as beneficiary and trustee of the Bellco Legacy Trust fbo Rebecka Belldegrun, (ii) 195,039 shares of common stock beneficially owned by Bellco Legacy LLC, of which Dr. Belldegrun is a manager, (iii) 1,798,163 shares of common stock beneficially owned by Vida Ventures LLC (“Vida”), of which VV Manager LLC is the manager, of which Dr. Belldegrun is a Senior Managing Director, (iv) 302,849 shares of common stock held by Dr. Belldegrun, 173,739 of which will be subject to our right of repurchase as of 60 days of March 31, 2022, and (v) 1,852,689 shares of common stock issuable upon exercise of options,1,640,103 of which will be unvested but exercisable within 60 days of March 31, 2022 held by Dr. Belldegrun. Dr. Belldegrun disclaims beneficial ownership of the shares held by Vida, except to the extent of any pecuniary interest therein. The address of Dr. Belldegrun, Bellco Legacy Trust and Bellco Legacy LLC is 2049 Century Park East, Suite 1940 Los Angeles, CA 90067. The address of Vida is 40 Broad Street, #201, Boston, MA 02109.
(5) Consists of (i) 2,186,198 shares of common stock held by David Chang, M.D., Ph.D., 10,191 of which will be subject to our right of repurchase as of 60 days of March 31, 2022, (ii) 1,201,108 shares of common stock held by the Chang 2006 Family Trust (“Chang Trust”), (iii) 856,044 shares of common stock held by the JEC 2019 Trust, (iv) 856,044 shares of common stock held by the RTC 2019 Trust, and (v) 3,328,238 shares of common stock issuable upon exercise of options, 2,718,587 of which will be unvested but exercisable within 60 days of March 31, 2022. Dr. Chang is co-trustee of the Chang Trust, JEC 2019 Trust and RTC 2019 Trust.
(6) Consists of 242,387 shares of common stock held by Joshua Kazam.
(7) Consists of (i) 131,216 shares of common stock held by Franz Humer, Ph.D. and (ii) 183,750 shares of common stock issuable upon exercise of options.
(8) Consists of (i) 6,535 shares of common stock held by the Messemer Family Trust (“Messemer Trust”) and (ii) 280,680 shares of common stock issuable upon exercise of options, 19,836 of which will be unvested but exercisable within 60 days of March 31, 2022. Ms. Messemer is trustee of the Messemer Trust.
(9) Consists of (i) 221,182 shares of common stock held by Owen Witte, M.D., and (ii) 54,040 shares of common stock issuable upon exercise of options, 2,335 of which will be unvested but exercisable within 60 days of March 31, 2022.
(10) Consists of 63,385 shares of common stock issuable upon exercise of options.
(11) Consists of 34,692 shares of common stock issuable upon exercise of options.
(12) Consists of (i) 54,466 shares of common stock held by Eric Schmidt, Ph.D., (ii) 1,152,595 shares of common stock held by the Eric Schmidt 2017 Family Irrevocable Trust (“Schmidt Trust”), which were acquired by the Schmidt Trust upon the exercise of stock options held by Dr. Schmidt, 40,707 of which will be subject to our right of repurchase as of 60 days of March 31, 2022, and (iii) 639,157 shares of common stock issuable upon exercise of options, 491,455 of which will be unvested but exercisable within 60 days of March 31, 2022. Dr. Schmidt’s spouse is trustee of the Schmidt Trust.
(13) Consists of (i) 77,675 shares of common stock held by Alison Moore, Ph.D., and (ii) 1,602,603 shares of common stock issuable upon exercise of options, 888,831 of which will be unvested but exercisable within 60 days of March 31, 2022.
(14) Consists of (i) 33,234 shares of common stock held by Rafael Amado, M.D., and (ii) 731,184 shares of common stock issuable upon exercise of options, 537,793 of which will be unvested but exercisable within 60 days of March 31, 2022.
(15) Consists of (i) 371,589 shares of common stock held by Veer Bhavnagri, and (ii) 645,904 shares of common stock issuable upon exercise of options, 394,382 of which will be unvested but exercisable within 60 days of March 31, 2022.
(16) Includes the shares described in notes (3) through (15) above.
| Trust is 811 Strada Vecchia Road, Los Angeles, CA 90077. The address of VVAG and Vida is 40 Broad Street, #201, Boston, MA 02109. The address of Bellco Capital LLC is 2049 Century Park East, Suite 1940 Los Angeles, CA 90067. This information is based on the Schedule 13G filed on February 13, 2019 with the SEC. |
(10) | Consists of (i) 74,870 shares of common stock held by Franz Humer, Ph.D. and (ii) 183,750 shares of common stock issuable upon exercise of options, 133,985 of which will be unvested but exercisable within 60 days of March 15, 2019 held by Dr. Humer.
|
(11) | Consists of (i) 37,432 shares of common stock held by Owen Witte, M.D. and (ii) 183,750 shares of common stock acquired by Dr. Witte upon the exercise of stock options, 133,985 of which will be subject to our right of repurchase as of 60 days of March 15, 2019.
|
(12) | Consists of the shares described in note (2) above.
|
(13) | Consists of (i) 6,535 shares of common stock held by the Messemer Family Trust (“Messemer Trust”) and (ii) 210,000 shares of common stock issuable upon exercise of options, all of which will be unvested but exercisable within 60 days of March 15, 2019 held by Debbie Messemer. Ms. Messemer is trustee of the Messemer Trust.
|
(14) | Includes the shares described in notes (5) through (13) above.
|
22
Delinquent Section
16(A) Beneficial Ownership Reporting Compliance16(a) Reports
Section 16(a) of the Exchange Act requires the Company’s directors and executive officers, and persons who own more than ten percent of a registered class of the Company’s equity securities, to file with the SEC initial reports of ownership and reports of changes in ownership of common stock and other equity securities of the Company. Officers, directors and greater than ten percent stockholders are required by SEC regulation to furnish the Company with copies of all Section 16(a) forms they file.
To the Company’s knowledge, based solely on a review of the copies of such reports furnished to the Company and written representations that no other reports were required, during the fiscal year ended December 31,
2018,2021, all Section 16(a) filing requirements applicable to its officers, directors and greater than ten percent beneficial owners were complied with,
except forother than one late
filing by Dr. Belldegrun as described below.On October 15, 2018, Vida purchased 194,445 shares of our common stock in connection with our initial public offering (the “Vida Purchase”). Dr. Belldegrun is a Senior Managing Director of VV Manager LLC, the manager of Vida, and may therefore be deemed to be the beneficial owner of the common shares held by Vida. The Vida Purchase was inadvertently omitted from Dr. Belldegrun’s Form 4 filedfiling reporting two transactions for Dr. Chang.
Executive Compensation
Compensation Discussion and Analysis
Overview
This Compensation Discussion and Analysis discusses the compensation philosophy, policies and principles underlying our executive compensation decisions for 2021. It provides qualitative information on
October 17, 2018. However, the
Form 4 was amended on February 13, 2019factors relevant to
includethese decisions and the
Vida Purchase.23
manner in which compensation is awarded to our named executive officers (our “Named Executive Compensation
Summary Compensation Table
The following table showsOfficers”) for the period from November 30, 2017 (inception) through December 31, 2017 and the fiscal year ended December 31, 2018, as applicable, compensation awarded to or paid to, or earned by, the Company’s named executive officers,2021, which consist of the individuals who served as the Company’sour principal executive officer, for any portion of 2018principal financial officer and the Company’s two other most highly compensatedour remaining three executive officers as of December 31, 2018, as follows:
during 2021. Our Named Executive Officers for 2021 were:•David Chang, M.D., Ph.D., our President and Chief Executive Officer;
Joshua Kazam, our former President;
•Eric Schmidt, Ph.D., our Chief Financial Officer;
•Rafael Amado, M.D., our Chief Medical Officer and Executive Vice President, Research and Development;
•Alison Moore, Ph.D., our Chief Technical Officer.
Officer; and2018
•Veer Bhavnagri, our General Counsel.
Executive Summary
2021 Business Highlights. During 2021, we achieved several important business milestones, including, but not limited to, the following:
•Advanced our innovative pipeline:
o Continued enrollment in the Phase 1 clinical trial ("ALPHA trial") of ALLO-501 in patients with relapsed/refractory non-Hodgkin lymphoma (“NHL”), the Phase 1/2 clinical trial ("ALPHA2 trial") of ALLO-501A in patients with relapsed/refractory NHL, and in the Phase 1 clinical trial ("UNIVERSAL trial") of ALLO-715 in patients with relapsed/refractory multiple myeloma. In addition, in the first half of 2021, we expanded the UNIVERSAL trial to assess ALLO-715 in combination with SpringWorks Therapeutics, Inc.’s investigational gamma secretase inhibitor, nirogacestat.
o Reported interim data for the ALPHA, ALPHA2 and UNIVERSAL trials at the American Society of Hematology ("ASH") annual meeting in December 2021, providing continued proof-of-concept of allogeneic chimeric antigen receptor (“CAR”) T cell therapy in blood cancers.
o Advanced our anti-CD70 CAR T cell product candidate, ALLO-316, that may have potential for various hematologic and solid tumor indications. In the first half of 2021, we initiated a Phase 1 clinical trial of ALLO-316 in patients with advanced or metastatic clear cell renal cell carcinoma.
o Progressed our technology platform that we call “TurboCARs” to mimic cytokine activation signaling within a CAR T cell, which could enhance the proliferative potential, migratory behavior, and killing activity of cells. In mid-2021, we initiated a Phase 1 clinical trial of our first TurboCAR, ALLO-605, in patients with relapsed/refractory multiple myeloma.
o Obtained FDA designations for advancing the development or any commercialization of our product candidates, including Fast-Track designation for ALLO-605, Regenerative Medicine Advanced Therapy designation for ALLO-715 and Orphan Drug Designation for ALLO-715.
•Further built-out our capabilities:
o Advanced Allogene Overland Biopharm (CY) Limited, our joint venture with Overland Pharmaceuticals (CY) Inc. for the development, manufacturing and commercialization of certain of our product candidates targeting BCMA, CD70, FLT3, and DLL3 in China, Taiwan, South Korea and Singapore. In 2021, we hired a new chief executive officer, leased a facility for manufacturing and began technology transfer for manufacturing.
o Initiated GMP manufacturing from our cell therapy manufacturing facility in Newark, California.
o Entered into a lease agreement to expand our headquarters in South San Francisco, California. The lease is expected to commence on April 1, 2022, and the additional 47,566 square feet of office and laboratory space will support the growth of our research platform and correlative clinical development activities.
o Strengthened our board of directors with the appointment of Ms. Barrett and Dr. Sato, and our scientific advisory board with the appointment of Jae Park, M.D.
o Grew our organization from 264 employees at the beginning of the year to 301 employees at the end of the year.
In addition to the above achievements, we overcame a significant unexpected regulatory challenge. In October 2021, the FDA placed a hold on our clinical trials. The clinical hold followed our notification to the FDA of a chromosomal abnormality in an ALPHA2 trial patient which was detected in a bone marrow biopsy undertaken to assess pancytopenia. Investigations concluded that the chromosomal abnormality was unrelated to TALEN gene editing or our manufacturing process and had no clinical significance. The investigation also determined that the abnormality was not detected in any of our manufactured product candidates or in any other patient treated with the same ALLO-501A lot. The abnormality occurred in the patient after the cell product was administered and involved regions of the T cell receptor and immunoglobulin genes known to undergo rearrangement as part of the T cell or B cell maturation process. The FDA found that we satisfactorily addressed all clinical hold issues and removed the hold in January 2022. The Compensation Table | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name and principal position | | Year | | | Salary
($) (1) | | | Stock
awards
($)(2) | | | Option
awards
($)(3) | | | Non-equity
incentive
plan
compensation
($)(4) | | | All other
compensation
($)(5) | | | Total
($) | |
David Chang, M.D., Ph.D. | | | 2018 | | | | 362,413 | | | | 8,948,152 | | | | 3,070,331 | | | | 217,641 | | | | 250,000 | | | | 12,848,537 | |
President and Chief Executive Officer | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | |
Joshua Kazam (6) | | | 2018 | | | | — | | | | 3,592,474 | | | | — | | | | — | | | | — | | | | 3,592,474 | |
Former President | | | 2017 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | |
| | | | | | | |
Eric Schmidt, Ph.D. | | | 2018 | | | | 201,232 | | | | — | | | | 2,299,658 | | | | 88,099 | | | | — | | | | 2,588,989 | |
Chief Financial Officer | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | |
Alison Moore, Ph.D. | | | 2018 | | | | 233,265 | | | | — | | | | 1,401,225 | | | | 102,124 | | | | 100,000 | | | | 1,836,614 | |
Chief Technical Officer | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
(1) | The dollar amounts in this column for Dr. Chang represent (i) $90,750 in consulting fees paid to Dr. Chang between April and June 2018 for services consistent with the role and duties of Chief Executive Officer and (ii) $271,663 in salary paid to Dr. Chang since the commencement of his employment in June 2018 (prorated based on an annual salary of $525,000). For more information, see description of our consulting arrangement with Dr. Chang below under “Transactions With Related Persons— Consulting Arrangements”. The dollar amounts in this column for Dr. Schmidt and Dr. Moore represent salary paid to each of Dr. Schmidt and Dr. Moore since the commencement of their employment (prorated based on an annual salary of $375,000 and $400,000, respectively).
|
(2) | In connection with the issuance of the Company’s Series A and SeriesA-1 convertible preferred stock in April 2018, the Company’s founders, including Dr. Chang and Mr. Kazam, agreed to modify their fully vested founders’ shares of common stock outstanding to include a forfeiture restriction that lapses based on their continued service to the Company. As such, the modified founders’ shares of common stock became compensatory upon such modification. The dollar amounts in this column represent the aggregate fair value (measured as of the modification date) of the modified shares beneficially owned by Dr. Chang and Mr. Kazam at the time of the modification. The dollar amounts in this column do not include an aggregate of $5,270,698 in fair value (measured as of the modification date) of modified shares that have a forfeiture restriction that lapses based on Mr. Kazam’s continued service to the Company. These shares have not been included in this column as they were not beneficially owned by Mr. Kazam at the time of the modification.
|
(3) | The dollar amounts in this column represent the aggregate grant date fair value of stock option awards granted in 2018. These amounts have been computed in accordance with FASB ASC Topic 718, using the
|
24
| Black-Scholes option pricing model. For a discussion of valuation assumptions, see Note 12 “Stock-based Compensation” to our financial statements included in our Annual Report on Form10-K for the year ended December 31, 2018. |
(4) | The dollar amounts in this column represent annual performance-based bonuses earned for 2018. For more information, see below under “Annual Performance-Based Bonus Opportunity.”
|
(5) | The dollar amounts in this column represent relocation bonuses paid to each of Dr. Chang and Dr. Moore.
|
(6) | Mr. Kazam resigned as our President on June 25, 2018.
|
Annual Base Salary
TheCommittee and the Board refrained from rewarding any additional compensation during the clinical hold and approved the 2021 annual incentive bonus and 2022 base salary adjustments after the clinical hold was resolved in January 2022.
We believe the achievements highlighted above as well as resolving the clinical hold were a result of exceptional strategic, technical and operational leadership.
2021 Executive Compensation Policies and Practices. During 2021, our executive compensation policies and practices included the following:
•Compensation Committee of Independent Directors. The Compensation Committee is composed of all independent directors and includes our lead independent director.
•Annual Compensation Review. The Compensation Committee undertakes a comprehensive review of compensation of our namedsenior vice presidents and above, including our Named Executive Officers, on an annual basis.
•Independent Compensation Consultant. The Compensation Committee engages its own compensation consultant, and reviews its independence from management.
•Risk Analysis. We review the structure of our executive compensation program to minimize the risk of inappropriate risk-taking by our executive officers.
•No Guaranteed Compensation. Although we have signed employment agreements with each of our Named Executive Officers, these agreements provide for “at will” employment, and none of these agreements provides any guarantees relating to base salary increases or the amounts of any annual incentive awards or long-term equity awards.
•Multi-Year Vesting. The equity awards granted to our executive officers generally vest over multi-year periods, consistent with current market practice and our retention objectives.
•No Special Retirement Benefits. We do not provide pension arrangements or post-retirement health coverage for our executive officers or employees. Our executive officers and other U.S.-based employees are eligible to participate in our Section 401(k) plan, which is a retirement savings defined contribution plan established in accordance with Section 401(a) of the Internal Revenue Code of 1986 (the “Code”). We currently make matching contributions into the Section 401(k) plan on behalf of participants. We match 100% of eligible contributions up to the first 3% of eligible compensation, with an additional match of 50% on the next 3% (maximum of 4.5%).
•No Special Health or Welfare Benefits or Perquisites. Our executive officers participate in broad-based company-sponsored health and welfare benefits programs on the same basis as our other full-time, salaried employees. We generally do not provide perquisites or other personal benefits to our executive officers other than those we provide to our employees generally.
•No Tax Reimbursements. We do not provide any tax reimbursement payments (including “gross-ups”) on any perquisites or other personal benefits.
•Policy Against Hedging and Speculative Trading and Pledging our Common Stock. Our insider trading policy prohibits our employees from engaging in “hedging” or other inherently speculative transactions with respect to our common stock or borrowing against our common stock. Specifically, no officer, director, other employee or consultant may engage in short sales, transactions in put or call options, hedging transactions or other inherently speculative transactions with respect to our
common stock at any time. In addition, no officer, director, other employee or consultant may margin, or make any offer to margin, or otherwise pledge as security, any of our common stock, including without limitation, borrowing against such stock, at any time.
•Stock Ownership Guidelines Policy. The purpose of our Stock Ownership Guidelines Policy is to encourage ownership of our common stock, promote the alignment of the long-term interests of our non-employee directors, Chief Executive Officer, and other executive officers with the long-term interests of our stockholders, and further promote our commitment to sound corporate governance. Under the Guidelines, the target stock ownership level for our President and Chief Executive Officer is six times (6x) his base annual salary, the target stock ownership level for our non-employee directors is five times (5x) their base annual cash retainer and the target stock ownership level for our other executive officers is generally set forth in each officer’sone times (1x) their base annual salary. Under these Guidelines, the compliance deadline for all of our executive officers and directors is the later of December 2023 and five years from an individual's commencement of employment letter agreement with us and periodically reviewed and adjusted as necessary byor appointment to our Board, although we expect that the target stock ownership levels likely will be achieved much sooner than that.
2021 Executive Compensation Highlights. Despite the challenge of Directors, based on the recommendation ofclinical hold, we made significant progress towards critical clinical, research, manufacturing, and other strategic milestones, as described above. With respect to 2021 compensation decisions, the Compensation Committee and our Board focused on ensuring that a significant portion of the total direct compensation awarded to our Named Executive Officers was performance-based and linked to meeting our long-term strategic plan to create long-term stockholder value.
The substantial majority of our Board2021 compensation to Named Executive Officers was in the form of Directors. In 2018, the base salaries forequity incentive awards. We believe that equity incentive awards further our named executive officers were $525,000, $375,000 and $400,000, for Dr. Chang, Dr. Schmidt and Dr. Moore, respectively. Joshua Kazam, our former President, did not receive a salary for 2017 or 2018.long-term strategic plan to create long-term stockholder value. In recognition of Dr. Chang’s, Dr. Schmidt’s and Dr. Moore’sour significant achievements and as a retention measure, the Compensation Committee also granted 2021 annual equity awards to Dr. Schmidt, Dr. Amado, Dr. Moore and Mr. Bhavnagri and our Board granted 2021 annual equity awards to Dr. Chang. The following charts illustrate the portion of compensation attributable to equity incentive awards, annual performance-based cash incentive awards and base salary for our Chief Executive Officer and for our other Named Executive Officers, as a group.
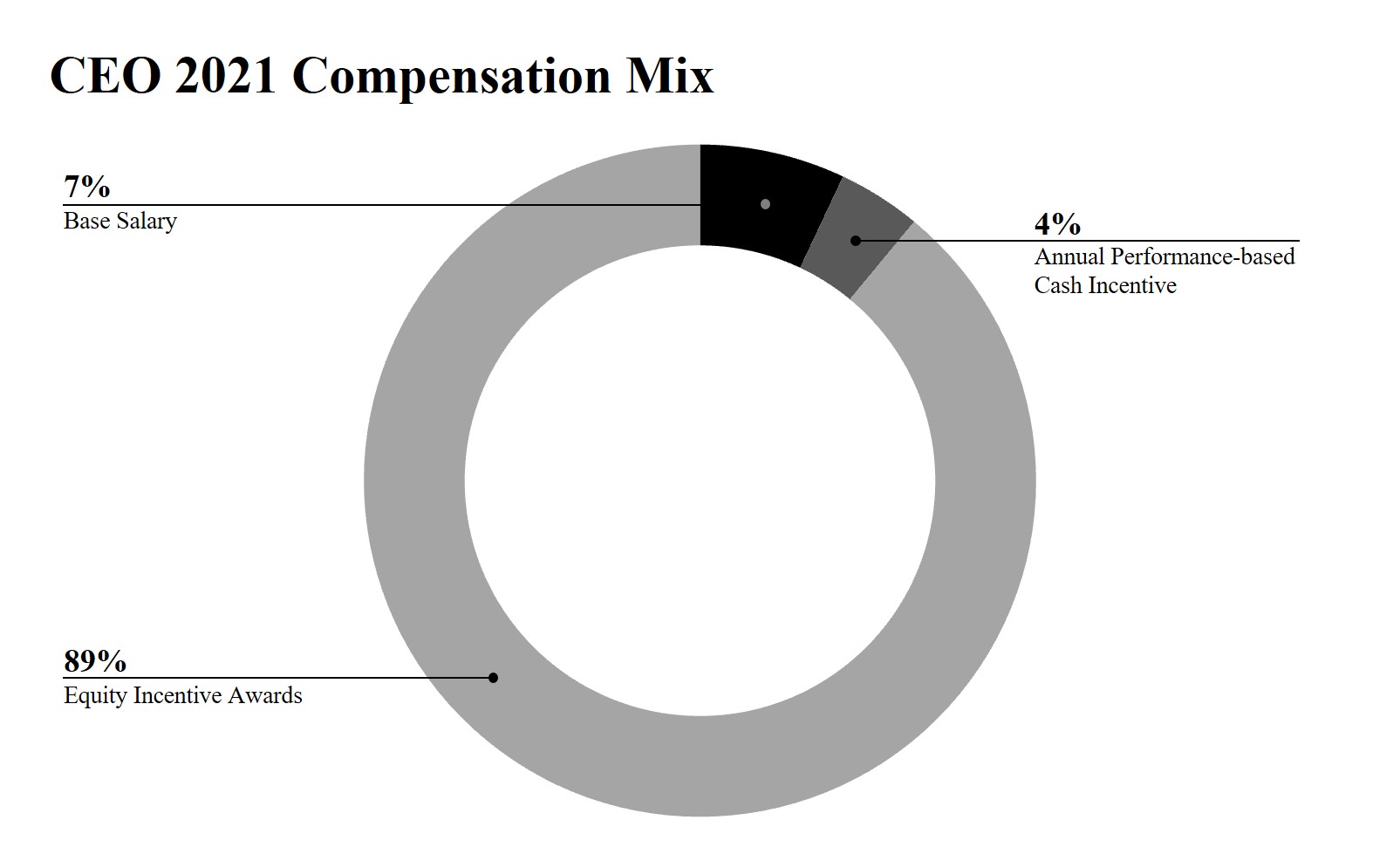
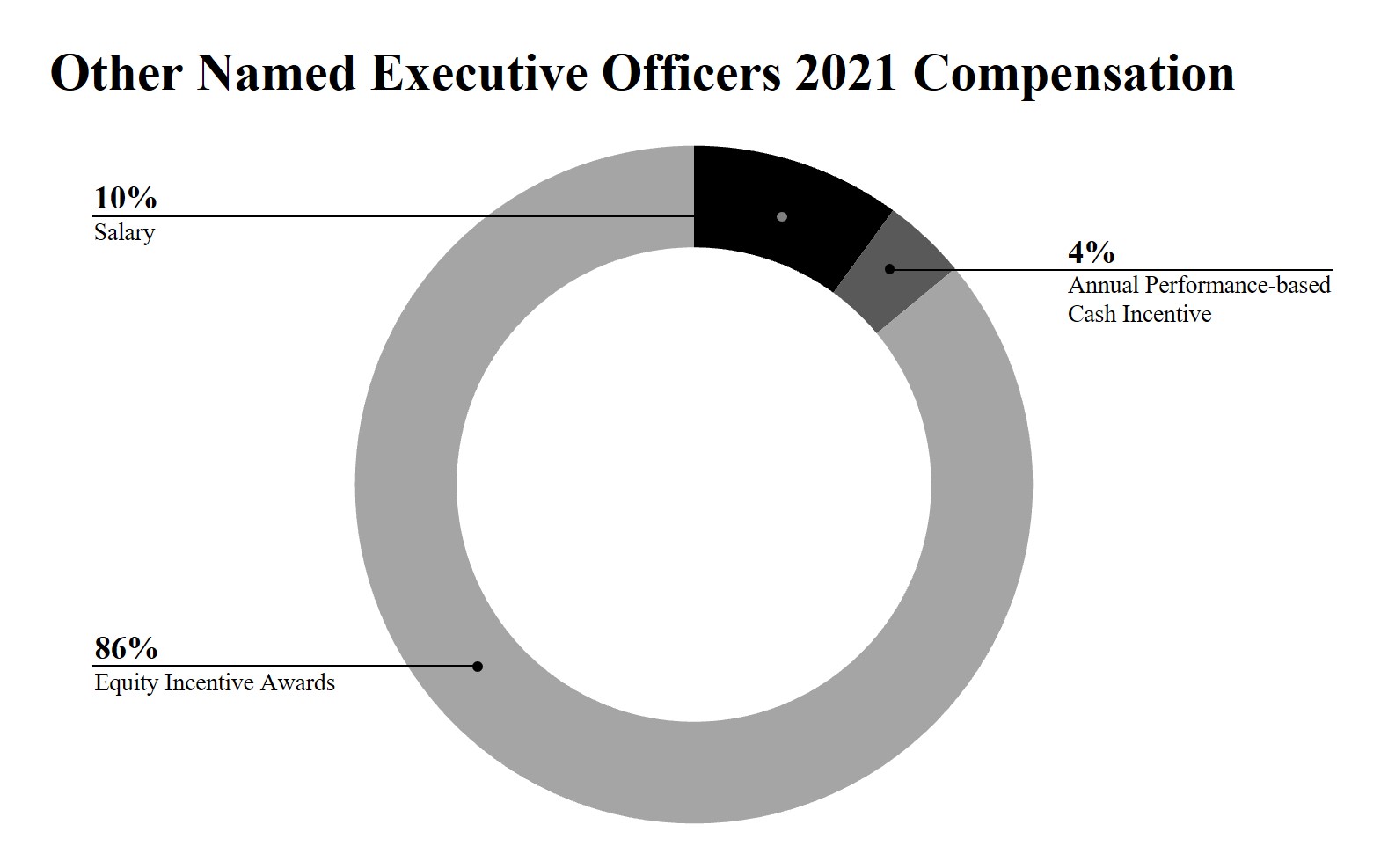
Executive Compensation Philosophy and Overview
Our executive compensation program is intended to meet five principal objectives:
•Enable us to attract, retain and motivate superior talent;
•Link rewards to the achievement of critical strategic priorities;
•Create incentives for our executive officers to further our long-term strategic plan to create long-term stockholder value;
•Provide appropriate levels of risk and reward relative to an executive officer's position with us; and
•Differentiate compensation based on individual performance.
Based on this philosophy, our performance-driven compensation program primarily consists of three components: base salary, annual cash bonus opportunity, and long-term incentive compensation in the form of equity awards. The Compensation Committee has determined that these three components, with a portion of target total direct compensation allocated to “at-risk” performance-based incentives through the use of short-term and long-term incentive compensation, best align the interests of our executive officers with those of our stockholders. While it does not have any formal policies for allocating compensation among the three components, the
Compensation Committee reviews relevant competitive market data and uses its judgment to determine the appropriate level and mix of compensation on an annual basis to ensure that compensation levels and opportunities are competitive and that we are able to attract and retain capable executive officers to work for our long-term prosperity and stockholder value, without taking unnecessary or excessive risks.
Process for Setting Executive Compensation
We seek to foster a performance-oriented culture, where individual performance is aligned with organizational objectives. To achieve this, we evaluate and reward our executive officers based on their contributions to the achievement of annual goals and objectives set early in the year. Performance is reviewed at least annually through processes discussed further below, with a focus on our research, clinical, regulatory, financial and operational performance, and in view of economic and financial conditions affecting the performance period. In addition, the Compensation Committee and our Board considered the positive results of the shareholder advisory vote on executive compensation held in 2021, where 69% of shareholders voted in favor of, and concluded our approach to compensation for named executive officers was appropriate.
Role of the Compensation Committee
The Compensation Committee reviews and approves our executive compensation philosophy, objectives and methods, evaluates our performance and the performance of our executive officers, and either approves executive compensation or makes recommendations for ratification by our Board members. Please see “Information Regarding Committees of our Board of Directors—Compensation Committee—Compensation Committee Processes and Procedures” for additional information.
Awards of performance-based compensation for the previous year are typically made at the last-scheduled Compensation Committee meeting of the year. Adjustments to the base salaries of our Named Executive Officers, if any, are also typically made at the last-scheduled Compensation Committee meeting of the year. Due to the clinical hold in the fourth quarter of 2021, our Board and Compensation Committee deferred year-end compensation matters until after resolution of the clinical hold in January 2022. The Compensation Committee typically reviews annual equity awards in the first quarter of the year.
Role of Management
In making compensation decisions, the Compensation Committee considers the recommendations of our Chief Executive Officer, with input from our Executive Chair, General Counsel and Secretary. Our Chief Executive Officer, Dr. Chang, makes recommendations to the Compensation Committee with respect to our executive officers, but is not present and does not participate in the deliberations or determination of his own compensation. The Compensation Committee may review and approve the corporate objectives and goals pursuant to the powers delegated under its charter. Dr. Chang annually leads the development of our corporate objectives and goals, which are typically reviewed and approved by the Compensation Committee and then our Board. Dr. Chang provided our business and operations perspective for the Compensation Committee’s final review of progress made on the goals set for 2021. Other than as described above, no other executive officers participate in the determination or recommendation of the amount or form of executive officer compensation.
Role of Compensation Consultant
The Compensation Committee is authorized to engage a compensation consultant or other advisors to review our executive officers’ compensation, including preparing an analysis against the compensation of executive officers at comparable companies, to ensure that our compensation is market competitive, with the goal of retaining and adequately motivating our senior management. During 2021, the Compensation Committee engaged Compensia to make recommendations for updating our compensation peer group, and to review and make recommendations regarding our equity plan and executive and director compensation for 2021. Compensia was invited to attend Compensation Committee meetings where they presented and discussed their analysis and findings. For 2021, with the assistance of Compensia, the Compensation Committee updated our compensation peer group, described below in the section entitled “Peer Companies and Market Compensation Data.”
In December 2018, December 2019, September 2020 and September 2021, the Compensation Committee analyzed whether the work of Compensia as a compensation consultant has raised any conflict of interest, taking into consideration the following factors: (i) the provision of other services to our Company by Compensia; (ii) the amount of fees from our Company paid to Compensia as a percentage of the firm’s total revenue; (iii) Compensia’s policies and procedures that are designed to prevent conflicts of interest; (iv) any business or personal relationship of Compensia or the individual compensation advisors employed by the firm with an executive officer of our Company; (v) any business or personal relationship of the individual compensation advisors with any member of the Compensation Committee; and (vi) any stock of our Company owned by the individual compensation advisors employed by Compensia. The Compensation Committee determined, based on its analysis of the above factors, that the work of Compensia and the individual compensation advisors employed by Compensia has not raised any conflict of interest and the Compensation Committee is satisfied with the independence of Compensia.
Peer Group and Market Compensation Data
When making compensation decisions, the Compensation Committee reviews the compensation of similarly-situated executive officers at companies that we consider to be our peers, taking into consideration the experience, position and functional role, level of responsibility and uniqueness of applicable skills of both our executive officers and those of our peers, and the demand and competitiveness for attracting and retaining an individual with each executive officer’s specific expertise and experience in the biotechnology industry. While this analysis is helpful in determining market-competitive compensation for senior management, leading to better attraction and retention of top-quality executive officers, it is only one factor in determining our executive officers’ compensation, and the Compensation Committee exercises its judgment in determining the nature and extent of its use.
With the assistance of its compensation consultant, and after taking into consideration such factors as product development stage, market capitalization, acquisitions, number of employees and public status, the following companies were selected and approved by the Compensation Committee in September 2020 to comprise our compensation peer group:
| | | | | | | | |
| Acceleron Pharma | Agios Pharmaceuticals | BeiGene |
| bluebird bio | Blueprint Medicines | CRISPR Therapeutics |
| Editas Medicine | Fate Therapeutics | Global Blood Therapeutics |
| Immunomedics | Iovance Biotherapeutics | Mirati Therapeutics |
| Moderna | MyoKardia | Nektar Therapeutics |
| Sage Therapeutics | Sarepta Therapeutics | Ultragenyx Pharmaceutical |
The Compensation Committee reviews the compensation peer group periodically to reflect changes in market capitalization and other factors, including number of employees, acquisitions and product development stage, and revises the companies included in the peer group accordingly. In this regard, the peer group was revised and approved by the Compensation Committee in September 2021 to consist of:
| | | | | | | | |
| Acceleron Pharma | Agios Pharmaceuticals | Beam Therapeutics |
| bluebird bio | CRISPR Therapeutics | Editas Medicine |
| Fate Therapeutics | Global Blood Therapeutics | IGM Biosciences |
| Instil Bio | Intellia Therapeutics | Iovance Biotherapeutics |
| Lyell Immunopharma | Mirati Therapeutics | Nektar Therapeutics |
| Rubius Therapeutics | Sana Biotechnology | Sorrento Therapeutics |
| SpringWorks Therapeutics | Turning Point Therapeutics | Ultragenyx Pharmaceutical |
| Xencor | | |
Executive Compensation Program and Compensation Decisions for the Named Executive Officers
The components of our executive compensation program in 2021 were as follows:
Annual Base Salary
The base salaries of our executive officers are designed to compensate them for day-to-day services rendered during the fiscal year. Appropriate base salaries are used to recognize the experience, skills, knowledge and responsibilities required of each executive officer and to allow us to attract and retain individuals capable of leading us to achieve our business goals in competitive market conditions.
The base salaries of our executive officers are reviewed at least annually by the Compensation Committee and adjustments are made to reflect Company and individual performance, as well as competitive market practices. The Compensation Committee also takes into account subjective performance criteria, such as an executive officer’s ability to lead, organize and motivate others, develop the skills necessary to mature with us, set realistic goals to be achieved in their respective area, and recognize and pursue new business opportunities that enhance our growth and success. The Compensation Committee does not apply specific formulas to determine increases, but instead makes an evaluation of each executive officer’s contribution to our long-term success. Annual adjustments to base salaries are effective as of January 1 of each year, with mid-year adjustments to base salaries made under special circumstances, such as promotions, retention measures or an increase in responsibilities.
The 2021 annual base salaries for our Named Executive Officers were as follows:
| | | | | | | | |
| Name | | 2021
Base Salary
($) |
| David Chang, M.D., Ph.D. | | 695,000 | |
| Eric Schmidt, Ph.D. | | 475,000 | |
| Rafael Amado, M.D. | | 600,000(1) |
| Alison Moore, Ph.D. | | 475,000 | |
| Veer Bhavnagri | | 464,000 | |
(1) Dr. Amado's salary was increased from $525,000 to $600,000 on May 18, 2021 as described below.
On May 18, 2021, the Compensation Committee reviewed the performance and compensation of Dr. Amado. To that date, Dr. Amado had several extraordinary achievements, including initiating our clinical trial of ALLO-316 in advanced or metastatic clear cell renal cell carcinoma, accelerating the advancement of our TurboCAR technology and achieving clearance of an IND for our first TurboCAR candidate, ALLO-605, in relapsed/refractory multiple myeloma, and advancing positive data from our clinical trials of ALLO-501 and ALLO-501A in relapsed/refractory non-Hodgkin lymphoma. In light of these achievements and as a retention measure, the Compensation Committee increased Dr. Amado’s annual base salary for the remainder of 2021 from $525,000 to $600,000.
After resolution of the clinical hold in January 2022, the Compensation Committee reviewed the annual base salary and target annual cash bonus opportunity of Dr. Chang, Dr. Amado, Dr. Schmidt, Dr. Moore and Mr. Bhavnagri. In recognition of achievements and as a retention measure, the Compensation Committee recommended our Board increase Dr. Chang’s annual base salary for
20192022 from $675,000 to
$600,000,$724,000, and
theincrease his target annual cash bonus opportunity from 60% of annual base salary to 65% of annual base salary for 2022. The Compensation Committee
also approved
anthe increase
of Dr. Amado's annual base salary for 2022 from $600,000 to
each of$615,000, Dr. Schmidt’s and Dr. Moore’s annual base salary for
20192022 from $475,000 to
$425,000.Annual Performance-Based Bonus Opportunity
$490,000, and Mr. Bhavnagri's annual base salary for 2022 from $464,000 to $480,000. In addition, the Compensation Committee increased Dr. Amado's, Dr. Schmidt's, Dr. Moore's and Mr. Bhavnagri's target annual cash bonus opportunity from 40% of annual base salary to 45% of annual base salaries,salary for 2022. On January 27, 2022, based on the Compensation Committee’s recommendation, our named executive officersBoard approved the increase to Dr. Chang’s annual base salary and target annual cash bonus opportunity.
Annual Cash Bonus Opportunity
Our Named Executive Officers are eligible to receive annual performance-based
cash bonuses, which are designed to provide appropriate incentives to our executive officers to achieve
pre-established annual corporate goals and to reward them for individual
achievementperformance towards these goals. The annual performance-based
cash bonus each
named executive officercurrent Named Executive Officer is eligible to receive is generally based on the extent to which we achieve the corporate goals that our Board
of Directors establishes each year. At the end of the year,
each of our Board
of Directors and
the Compensation Committee
reviewsreview our performance
against each corporate goal and
approvesapprove the extent to which we achieved each of
ourthese annual corporate goals.
Our Generally, our Board of Directors and the Compensation Committee, as applicable, will generally considerassess each named executive officer’sNamed Executive Officer’s individual contributions towards reaching our annual corporate goals but does not typically establish specific individual goals for our named executive officers. There is no minimumNamed Executive Officers.
For 2021, the target annual cash bonus
percentage or amount established for the named executive officers and, thus, the bonus amounts vary from year to year based on corporate and individual performance. For 2018,opportunity of Dr. Chang was
eligible to receive a target bonus of up to 45%set at 60% of his
annual base salary
proratedand the target annual cash bonus opportunities for
the partial yeareach of
service, pursuant to the terms of his employment letter agreement described below. For 2018,Dr. Amado, Dr. Schmidt,
was eligible to receive a target bonus of up to 35% of his base salary, prorated for the partial year of service, pursuant to the terms of his employment letter agreement described below. For 2018, Dr. Moore
and Mr. Bhavnagri was
eligibleset at 40% of each Named Executive Officer's annual base salary. Our Board and the Compensation Committee may award above-target bonuses, which was limited in 2021 to
receive a130% of the target
annual cash bonus
of up to 35% of her base salary, proratedopportunities, for
the partial year of service, pursuant to the terms of her employment letter agreement described below.extraordinary performance.
The corporate goals used in our 2021 annual cash bonus plan were proposed by management, and reviewed and approved by the Compensation Committee and our Board. Our Board considered and assigned a relative weight to each corporate goal to appropriately focus efforts on achievements that were intended to enhance stockholder value.
Our core corporate goals for 2021 and the relative weighting established by our Board of Directors for 2018 consistedeach corporate goal were as follows:
•Deliver Phase 1 data and initiate the Phase 2 portion of the following:Company StandupALPHA2 trial of ALLO-501A by the end of 2021 (weighted at 40%)
; Onboard employees conveyed as part•Deliver data for a Phase 2 decision for ALLO-715 with or without nirogacestat by the end of the acquisition of allogeneic chimeric antigen receptor (“CAR”) T cell assets from Pfizer, Inc. in April 2018;
Implement key human resources and finance systems; and
Execute a lease for securing Company headquarters.
25
Submit Anti-CD52 Monoclonal Antibody Drug Master File with the U.S. Food and Drug Administration (“FDA”)2021 (weighted at 20%)
Achieve;
ALLO-501• Investigational New Drug Application (“IND”) ReadinessObtain IND clearance for a First Quarter 2019 IND Submission toPhase 1 clinical trial of ALLO-605 by the FDAend of the second quarter of 2021 (weighted at 10%);
•Generate interim data from the Phase 1 clinical trial of ALLO-316 by the end of 2021 (weighted at 20%); and Complete•Produce a clinical GMP batch at our manufacturing facility in Newark, California by the majorityend of IND modules;
CompleteGMP-based manufacturing runs; and
Initiate clinical sitestart-up activities.
AchieveALLO-715 IND Readiness for a 2019 IND Submission to the FDA2021 (weighted at 20%10%)
.Complete pilot production; and
Convene a multiple myeloma clinical scientific advisory board.
TheOur Board also established an additional, or stretch, corporate goalgoals to complete by the end of 2021 as follows:
•Nominate a second roundnext-generation product candidate (weighted at 10%);
•Complete dosing of capital raising, weighedthe first two cohorts in the Phase 1 clinical trial of ALLO-605 (weighted at 25%.No specific individual goals were established for any10%); and
•Complete the planned dose escalation of our named executive officers for 2018.the Phase 1 clinical trial of ALLO-316 (weighted at 10%).
In December 2018,2021, our Board of Directorsreviewed our 2021 corporate goals and gave feedback to management and the Compensation Committee determined thatfor further discussion and review in January 2022. Our Board and the 2018Compensation Committee did not approve any annual bonus payment prior to the resolution of the clinical hold in early January 2022.
After the resolution of the clinical hold, the Compensation Committee and our Board awarded partial (20%) credit to the first corporate goal, as we reported interim clinical data from ALPHA2 at the ASH annual meeting in December 2021, but did not initiate the Phase 2 trial. They also awarded partial (10%) credit to the second corporate goal as we finished dosing an initial cohort of patients to assess the ALLO-715 and nirogacestat combination, but we are following the patients prior to any decision on further enrollment or initiation of Phase 2. The Compensation Committee and our Board awarded full credit to the remaining corporate goals includingas all had been achieved. With respect to the stretch corporate goal, had been achieved at an aggregate level of 125%. The Board andgoals, the Compensation Committee also noted thatand our Board awarded credit to the first stretch goal, but no credit to the other two stretch goals, which were not achieved.
In addition, the Compensation Committee and our Board awarded an additional 15% of credit for our perseverance in addition to meetingovercoming the corporate goals,clinical hold and certain additional accomplishments, including:
•Completed the Company achieved significant additional milestones, includingfirst dose cohort in the ALLO-605 phase 1 study;
•Manufactured not just one, but not limited to completing boththree, clinical GMP batches at our manufacturing facility;
•Hired a convertible note round of financing as well as completingVice President, Commercial, and advanced an initial public offering, executing a leasecommercial strategy and roadmap;
•Executed on strategic business development and research initiatives; and
•Secured additional lab and office space at our headquarters to secure additional laboratory space, initiating clinical sitestart-upaccommodate future company growth. activities for theALLO-501 clinical trial, and assembling an exceptional senior leadership team. As a result,
in December 2018,the Compensation Committee approved awarding Dr. Schmidt, Dr. Amado, Dr. Moore and Mr. Bhavnagri 95% of their respective target annual cash bonus opportunity for 2021, and recommended and our Board
approved awarding Dr. Chang 95% of
Directors awarded the followinghis target annual cash bonus
opportunity for 2021. In addition, due to Dr.
ChangAmado’s and
ourDr. Moore's exceptional scientific performance and leadership in overcoming the clinical hold in a timely and complete manner, the Compensation Committee awarded
the following bonuses to Dr.
SchmidtAmado a separate discretionary bonus of $100,000 and Dr.
Moore: | | | | | | |
Executive Officer | | Title | | 2018
Bonus
Amount(1) | |
David Chang, M.D., Ph.D. | | President and Chief Executive Officer | | $ | 217,641 | |
Eric Schmidt, Ph.D. | | Chief Financial Officer | | $ | 88,099 | |
Alison Moore, Ph.D. | | Chief Technical Officer | | $ | 102,124 | |
(1) | As noted above, the 2018 bonus amounts are prorated for the partial year of service. Dr.
| | | | | | | |
| Name | | 2021
Annual Cash Bonus
($) |
David Chang, began his service with the Company in April 2018, and Dr.M.D., Ph.D. | | 396,150 | |
Eric Schmidt, and Dr.Ph.D. | | 180,500 | |
| Rafael Amado, M.D. | | 328,000 | |
Alison Moore, began their service with the Company in June 2018.Ph.D. | | 255,500 | |
| Veer Bhavnagri | | 176,320 | |
Mr. Kazam was not entitled
Long-Term Incentive Compensation
We provide long-term incentive compensation to any target or minimum bonus and no specific performance goals or bonus program were establishedour executive officers through the grant of equity awards. We believe that equity awards create incentives for our executive officers to further our long-term strategic plan to create long-term stockholder value. We also believe equity awards create an ownership culture. In addition, the vesting requirements of our equity awards contributes to executive retention by providing an incentive to our executive officers to remain employed by us during the vesting period.
Generally, significant equity awards are granted at the time
he served as an executive officer
of the Company.Equity-Based Incentive Awards
Our equity-based incentive awards are designed to align our interests with those of our employees and consultants, including our named executive officers. Our Board of Directors or any authorized committee thereof is responsible for approving equity awards, which include to date, stock options and restricted stock unit, or RSU, awards. Vesting of the stock option and RSU awards is tied to continuous service with us and serves as an
26
additional retention measure. Our executive officers generally were awarded an initial stock option grant upon commencement ofcommences employment. AdditionalThereafter, equity awards may be granted periodicallyat varying times and in varying amounts in the discretion of the Compensation Committee or, if awards are being
granted to specifically incentivizeour Chief Executive Officer, in the discretion of our Board, but are generally made once a year unless such executive officer is promoted, or for recognition of outstanding performance. None of our executive officers is currently party to an employment agreement that provides for an automatic grant of stock options or other equity awards.
Historically, we have granted equity awards to our executive officers in the form of options to purchase shares of our common stock. In March 2019, the Compensation Committee determined that an increasing number of companies in our compensation peer group grant full value awards, such as restricted stock unit ("RSU") awards and, therefore, decided that RSU awards should be added to our executive compensation program to attract and retain highly qualified executive officers. In this regard, while both stock options and RSU awards enable our executive officers to achieve certain corporate goals or to reward them for exceptional performance. As of December 31, 2018, stock option awards werebenefit, like stockholders, from any increases in the only form of equity awards we had granted to anyvalue of our executive officers.common stock, stock options deliver future value only if the value of our common stock increases above the exercise price. In March 2019, both stock option awards andcontrast, RSU awards were granteddeliver fully paid shares of our stock upon vesting, so, during periods of stock market volatility, RSU awards help retain employees. In addition, full value awards, such as RSU awards, are less dilutive to our named executive officers as partexisting stockholders since fewer shares are needed to achieve an equivalent value relative to stock options.
The exercise price of an annual grant program that applied to all employees.Prior to our initial public offering, we granted all equity awards pursuant to our prior amended and restated 2018 equity incentive plan (the “Prior Plan”). All equity awards granted since our initial public offering have been granted pursuant to our amended and restated 2018 Equity Incentive Plan (the “2018 Plan”), the terms of which are described below under “-Equity Benefit Plans.” All stock options are granted with a per share exercise priceis equal to no less than the fair market value of a share(our closing market price on the Nasdaq Global Select Market) of our common stock on the date of grant. Our stock options generally vest as to 1/4
th of the grant of such award.Generally, our stock option awards vest over a one to four-year periodshares subject to the holder’s continuous serviceoption upon the first anniversary of the grant date and 1/36
th of the remaining shares each month thereafter until such award is fully vested on the four year anniversary of the grant date, subject to usvesting acceleration as described under the heading “—Potential Payments Upon Termination or Change of Control” below. The RSU awards granted to our Named Executive Officers in 2021 generally vest in four equal annual installments from the grant date, subject to vesting acceleration as described under the heading “—Potential Payments Upon Termination or Change of Control” below. The other terms of the equity awards are governed by our 2018 Equity Incentive Plan, as amended (the “EIP”). We have granted and may becontinue to grant RSU awards that vest upon performance conditions to select executive officers in order to incentivize meeting specific strategic goals. In August 2019, the Compensation Committee granted with an early exercise feature. Such early exercise feature allows the holder to exercise and receive unvestedDr. Amado a new hire performance RSU award for 57,361 shares of our common stock, sowhich vests and settles in three equal installments, with (1) the first installment to vest upon initiation of a Phase 2 clinical trial of our first anti-CD19 product candidate, (2) the second installment to vest upon the filing of a biologics license application with the FDA for our first anti-CD19 product candidate, and (3) the third installment to vest upon FDA approval of our first anti-CD19 product candidate. Any unvested portion of the performance RSU award will expire after seven years.
The vesting schedule and the number of shares of our common stock granted are established to ensure a meaningful incentive to remain in our employ. Accordingly, the stock award will provide a return to the employee only if the employee remains in our service, and, in the case of the stock option component, only if the market price of our common stock appreciates over the option term. The Compensation Committee believes that the holdernew hire equity awards granted to Dr. Amado were necessary to secure his employment and to incentivize his long-term commitment, performance and support of our anti-CD19 product candidate, which is critical for our success.
In March 2021, the Compensation Committee granted our Named Executive Officers, and recommended a grant to our Chief Executive Officer that was approved by our Board, options to purchase shares of our common stock and RSU awards that may be settled for shares of our common stock over a four year time-based vesting schedule described above. In determining the number of shares of our common stock subject to the stock options and RSU awards granted to the Named Executive Officers, the Compensation Committee first determined the value of overall annual equity awards, and then determined the appropriate award mix. In March 2021, the Compensation Committee chose to deliver a value mix of approximately 70% stock options and approximately 30% RSUs, based on the accounting “fair value” as reflected in our Summary Compensation Table. The ratio of RSUs to stock options was based in part on an analysis of competitive market data prepared by the Compensation Committee’s compensation consultant and the Compensation Committee’s desire to tie a majority of the equity compensation program to stockholder value creation with stock options. The Compensation Committee also desired to provide a minority portion of the equity program with a durable retention profile, and the grant of RSU awards assists in retaining the team through periods of stock price volatility. The Compensation Committee also considered the equity awards granted at the 75th percentile to the executives holding comparable positions at the companies in our compensation peer group, as well as each Named Executive Officer’s existing equity holdings, level of responsibility and criticality, unvested status of existing equity holdings, and its subjective assessment of each Named Executive Officer’s individual performance and our overall company performance.
In addition, on May 18, 2021, the Compensation Committee reviewed the performance and compensation of Dr. Amado. To that date, Dr. Amado had several extraordinary achievements, including initiating our clinical trial of ALLO-316 in advanced or metastatic clear cell renal cell carcinoma, accelerating the advancement of our TurboCAR technology and achieving clearance of an IND for our first TurboCAR candidate, ALLO-605, in relapsed/refractory multiple myeloma, and advancing positive data from our clinical trials of ALLO-501 and ALLO-501A in relapsed/refractory non-Hodgkin lymphoma. In light of these achievements and as a retention measure, the Compensation Committee granted an RSU award that may vest and be settled for 200,534 units, with 33,422 vesting in
one year, 66,844 vesting in two years and 100,268 vesting in three years. Each unit granted pursuant to the RSU award represents a contingent right to receive one share of our common stock for each unit that vests. The Compensation Committee viewed the equity award as a critical retention measure ahead of our plans for rapid advancement of our pipeline, including the potential initiation of a planned pivotal trial of ALLO-501A and potential future submission for approval of ALLO-501A.
The equity awards granted to our Named Executive Officers during 2021 were as follows:
| | | | | | | | | | | | | | | | | | | | |
| Name | | Options to Purchase Shares of Our Common Stock (#) | | RSU Awards for Shares of Our Common Stock (#) | | Equity Awards (Aggregate Grant Date Fair Value) |
| David Chang, M.D., Ph.D. | | 298,836 | | 80,452 | | $9,070,304 |
| Eric Schmidt, Ph.D. | | 92,365 | | 24,866 | | $2,803,462 |
| Rafael Amado, M.D. | | 92,365 | | 225,400 | | $8,803,440 |
| Alison Moore, Ph.D. | | 92,365 | | 24,866 | | $2,803,462 |
| Veer Bhavnagri | | 82,469 | | 22,202 | | $2,503,104 |
Health and Welfare Benefits
Our Named Executive Officers, during their employment with us, are eligible to participate in our employee benefit plans, including our medical, dental, group term life, disability and accidental death and dismemberment insurance plans, in each case on the same basis as all of our other employees. In addition, we provide a Section 401(k) plan to our employees, including our Named Executive Officers, as discussed in the section below entitled “—Section 401(k) Plan.”
Section 401(k) Plan
We maintain a defined contribution employee retirement savings plan (the “401(k) Plan”), for our employees. Our executive officers are eligible to participate in the 401(k) Plan on the same basis as our other employees. The 401(k) Plan is intended to qualify as a tax-qualified plan under Section 401(a) of the Code. The 401(k) Plan provides that each participant may contribute up to the lesser of 100% of compensation or the statutory limit, which was $19,500 for calendar year 2021. Participants that are 50 years or older can also make “catch-up” contributions, which in calendar year 2021 may have a greater opportunity for gainsbeen up to an additional $6,500 above the statutory limit. We currently make matching contributions into the 401(k) Plan on behalf of participants. We match 100% of eligible contributions up to the first 3% of eligible compensation, with an additional match of 50% on the next 3% (maximum of 4.5%). Participant contributions are held and invested, pursuant to the participant’s instructions, by the plan’s trustee.
Perquisites and Other Personal Benefits
We generally do not provide perquisites or personal benefits to our Named Executive Officers. We do, however, pay a portion of the premiums for medical, dental, group term life, disability and accidental death and dismemberment insurance for all of our full-time employees. Our Board may elect to adopt qualified or nonqualified benefit plans in the future if it determines that doing so is in our best interests.
In 2020, we paid $83,256 to cover the Hart-Scott-Rodino (“HSR”) filing fees for Dr. Chang, which included a tax “gross-up” payment to cover the imputed income associated with this payment. The filing and the associated filing fee were triggered by regulatory requirements because Dr. Chang's total holdings of our common stock were above a specified value. Our Compensation Committee and Board determined it appropriate to pay the HSR filing fees and associated taxes that arose solely as a result of the price appreciation in shares accumulated by Dr. Chang over a long period of time during which he had made substantial contributions toward such appreciation, and recognizing that our Board and Compensation Committee encourages him to hold our shares for alignment with stockholders’ interests. Our Board and Compensation Committee considers this reimbursement to be taxedbusiness related for the above reasons, even though the HSR filing fee is personally attributable to the individual executive. No HSR filing fees were due or paid in 2021.
Post-Employment Compensation
Our Named Executive Officers are entitled to certain severance and change of control payments and benefits pursuant to our change in control and severance benefit plan (the “Plan”), as described in more detail below in the section entitled “—Potential Payments Upon Termination or Change of Control.” The Plan provides for a combination of a lump-sum cash severance payment, continued health benefits and acceleration of vesting on outstanding equity awards in specified circumstances. Acceleration of vesting is subject to a “double trigger” arrangement, meaning that vesting acceleration occurs only in the event of a change of control of the Company in connection with or followed by a termination of employment without cause by us, or with good reason by the Named Executive Officer.
Given the industry in which we participate and the range of strategic initiatives that we may explore, we believe these arrangements are an essential element of our executive compensation program and assist us in recruiting and retaining talented individuals. In addition, since we believe it may be difficult for our executive officers to find comparable employment following an involuntary termination of employment in connection with or following a change of control of the Company, these payments and benefits are intended to ease the consequences to an executive officer of an unexpected termination of employment. By establishing these payments and benefits, we believe we can mitigate the distraction and loss of executive officers that may occur in connection with rumored or actual fundamental corporate changes and thereby protect stockholder interests while a transaction is under consideration or pending.
Accounting and Tax Considerations
Under Financial Accounting Standard Board ASC Topic 718, or ASC Topic 718, we are required to estimate and record an expense for each share-based payment award (including stock options) over the vesting period of the award. We record share-based compensation expense on an ongoing basis according to ASC Topic 718. The Compensation Committee has considered, and may in the future consider, the grant of performance-based or other types of stock awards to our executive officers in lieu of or in addition to stock options and RSU awards in light of the accounting impact of ASC Topic 718 and other considerations.
Under Section 162(m) of the Internal Revenue Code (“Section 162(m)”), compensation paid to any publicly held corporation’s “covered employees” that exceeds $1 million per taxable year for any covered employee is generally non-deductible. However, Section 162(m) provides a reliance period exception for corporations that became publicly held before December 20, 2019, pursuant to which the deduction limit under Section 162(m) does not apply to certain compensation paid (or in some cases, granted) pursuant to a plan or agreement that existed during the period in which the corporation was not publicly held, subject to certain requirements and limitations. Under Section 162(m), this reliance period ends upon the earliest of the following: (i) the expiration of the plan or agreement; (ii) the material modification of the plan or agreement; (iii) the issuance of all employer stock and other compensation that has been allocated under the plan; or (iv) the first meeting of stockholders at which directors are to be elected that occurs after the close of the third calendar year following the calendar year in which the corporation’s initial public offering occurs. However, the reliance period exception under Section 162(m) may be repealed or modified in the future as a result of certain changes that were made to Section 162(m) pursuant to the Tax Cuts and Jobs Act.
Compensation paid to each of the Company’s “covered employees” in excess of $1 million per taxable year generally will not be deductible unless it qualifies for the reliance period exception under Section 162(m). Because of certain ambiguities and uncertainties as to the application and interpretation of Section 162(m), as well as other factors beyond the control of the Compensation Committee, no assurance can be given that any compensation paid by the Company will qualify for the reliance period exception under Section 162(m) and be deductible by the Company in the future. Although the Compensation Committee will continue to consider tax implications as one factor in determining executive compensation, the Compensation Committee also looks at other factors in making its decisions and retains the flexibility to provide compensation for our Named Executive Officers in a manner consistent with the goals of our executive compensation program and the best interests of the Company and its stockholders, which may include providing for compensation that is not deductible by the Company due to the deduction limit under Section 162(m). The Compensation Committee also retains the flexibility to modify compensation that was initially intended to be exempt from the deduction limit under Section 162(m) if it determines that such modifications are consistent with the Company’s business needs.
Risk Assessment Concerning Compensation Practices and Policies
The Compensation Committee annually reviews our compensation policies and practices to assess whether they encourage our employees to take inappropriate risks. After reviewing each of our compensation plans, and the checks and balances built into, and oversight of, each plan, in January 2022 the Compensation Committee determined that any risks arising from our compensation policies and practices for our employees are not reasonably likely to have a material adverse effect on our Company as a whole. In addition, the Compensation Committee believes that the mix and design of the elements of executive compensation do not encourage management to assume excessive risks and, as described above under the heading “Compensation Discussion and Analysis,” significant compensation decisions, and decisions concerning the compensation of our executive officers, include subjective considerations by the Compensation Committee or our Board, which restrain the influence of formulae or objective factors on excessive risk taking. Finally, the mix of short-term compensation (in the form of base salary and annual cash bonus, if any), and long-term capital gains rates rather than ordinary income rates. Fromincentive compensation (in the form of stock options and RSU awards) also prevents undue focus on short-term results and helps align the interests of our executive officers with the interests of our stockholders.
Compensation Committee Interlocks and Insider Participation
As noted above, the Compensation Committee consists of David Bonderman, Franz B. Humer and John DeYoung. None of the members of the Compensation Committee during 2021 has at any time been our officer or employee. None of the members of the Compensation Committee during 2021 had a relationship that must be described under the SEC rules relating to timedisclosure of related
person transactions. None of our executive officers serve, or in the past fiscal year has served, as a member of the board of directors or the compensation committee of any entity that has one or more of its executive officers serving on our Board of Directors or Compensation Committee.
Compensation Committee considers appropriate, we mayReport*
The Compensation Committee has reviewed and discussed the Compensation Discussion and Analysis required by Item 402(b) of Regulation S-K with management and, based on such review and discussion, the Compensation Committee recommended to our Board of Directors that the Compensation Discussion and Analysis be included in this Proxy Statement and incorporated into the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2021.
| | | | | | | | |
| | |
| | Compensation Committee |
| | David Bonderman, Chair |
| | Franz B. Humer |
| | John DeYoung |
| | | | | |
| * | The material in this report is not “soliciting material,” is not deemed “filed” with the SEC and is not to be incorporated by reference in any filing of the Company under the Securities Act or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing. |
2021 SUMMARY COMPENSATION TABLE
The following table sets forth all of the compensation awarded to, earned by or paid to our Named Executive Officers during the fiscal year ended December 31, 2021 and, with respect to Mr. Bhavnagri, during the fiscal year ended December 31, 2020, and, with respect to Dr. Chang, Dr. Schmidt, Dr. Amado and Dr. Moore, during the fiscal years ended December 31, 2020 and December 31, 2019.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name and principal position | | Year | | Salary ($)(1) | | Bonus ($) | | Stock awards ($)(2) | | Option awards ($)(3) | | Non-equity incentive plan compensation ($)(4) | | All other compensation ($)(5) | | Total ($) |
| David Chang, M.D., Ph.D. | | 2021 | | 695,000 | | — | | 2,717,669 | | 6,352,635 | | 396,150 | | — | | 10,161,454 |
| President and Chief Executive Officer | | 2020 | | 675,000 | | — | | — | | 6,550,535 | | 506,250 | | 83,256 | | 7,815,041 |
| | 2019 | | 600,000 | | — | | 4,651,970 | | 5,327,140 | | 363,000 | | 250,000 | | 11,192,110 |
| Eric Schmidt, Ph.D. | | 2021 | | 475,000 | | | | 839,973 | | 1,963,489 | | 180,500 | | — | | 3,458,962 |
| Chief Financial Officer | | 2020 | | 440,000 | | — | | 561,176 | | 1,262,643 | | 220,000 | | — | | 2,483,819 |
| | 2019 | | 425,000 | | — | | 1,328,366 | | 1,519,700 | | 187,000 | | — | | 3,460,066 |
| Rafael Amado, M.D. | | 2021 | | 571,591 | | 100,000 | | 6,839,951 | | 1,963,489 | | 228,000 | | 11,125 | | 9,714,156 |
| Chief Medical Officer and Executive Vice President of Research and Development | | 2020 | | 510,000 | | 75,000 | | 561,176 | | 1,262,643 | | 255,000 | | — | | 2,663,819 |
| | 2019 | | 145,833 | | — | | 4,249,976 | | 2,759,623 | | 71,726 | | 225,000 | | 7,452,158 |
| Alison Moore, Ph.D. | | 2021 | | 475,000 | | 75,000 | | 839,973 | | 1,963,489 | | 180,500 | | 13,050 | | 3,547,012 |
| Chief Technical Officer | | 2020 | | 440,000 | | — | | 561,176 | | 1,262,643 | | 220,000 | | — | | 2,483,819 |
| | 2019 | | 425,000 | | — | | 996,275 | | 1,519,700 | | 187,000 | | — | | 3,127,975 |
| Veer Bhavnagri | | 2021 | | 464,000 | | | | 749,984 | | 1,753,120 | | 176,320 | | 13,050 | | 3,156,474 |
| General Counsel | | 2020 | | 425,000 | | — | | 537,490 | | 1,211,953 | | 212,500 | | 9,385 | | 2,396,328 |
(1) The dollar amount reported for 2019 in this column for Dr. Amado represents salary paid to Dr. Amado since the commencement of his employment (prorated based on an annual salary of $500,000).
(2) The dollar amounts reported for 2019 and 2020 in this column reflect the aggregate grant date fair value of restricted stock units granted during 2019 and 2020 based on the closing market price of the Company's common stock on the date of grant.
(3) The dollar amounts in this column represent the aggregate grant date fair value of stock option awards granted in 2019, 2020 and 2021. These amounts have been computed in accordance with FASB ASC Topic 718, using the Black-Scholes option pricing model. For a discussion of valuation assumptions, see Note 10 “Stock-based Compensation” to our financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2021. These amounts do not reflect the actual economic value that will be realized by the Named Executive Officer upon the vesting of the stock options, the exercise of the stock options, or RSUthe sale of the common stock underlying such stock options.
(4) The dollar amounts in this column represent annual performance-based bonuses earned for 2019, 2020 and 2021. For more information, see above under “Annual Bonus Opportunity.”
(5) The dollar amounts in this column represent HSR filing fee and related tax gross-up to Dr. Chang in 2020, Section 401(k) Company contributions for Mr. Bhavnagri in 2021 and 2020, Section 401(k) Company contributions for Dr. Amado and Dr. Moore in 2021, relocation bonuses paid to Dr. Chang in 2019 and sign-on and relocation bonuses paid to Dr. Amado in 2019.
2021 GRANTS OF PLAN-BASED AWARDS TABLE
The following table sets forth information relating to the grant of plan-based incentive awards that vest upon achievement of performance goals.Agreements withto our Named Executive Officers
in 2021.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | Estimated Future Payouts Under Non-Equity Incentive Plan Awards(1) | | All Other Stock Awards: Number of Shares of Stock or Units (#) | | All Other Option Awards: Number of Securities Underlying Options (#) | | Exercise or Base Price of Option Awards
($/Sh) | | Grant Date Fair Value of Stock and Option Awards
($) |
| Name | | Grant Date
| | Threshold
($) | | Target
($) | | Maximum
($) | | | | |
| David Chang, M.D., Ph.D. | | | | | | | | | | | | | | | | |
| Restricted stock unit award | | 3/26/2021 | | — | | | — | | | — | | | 80,452 | | | — | | | — | | | 2,717,669 | |
| Stock option award | | 3/26/2021 | | — | | | — | | | — | | | — | | | 298,836 | | | 33.78 | | | 6,352,635 | |
| Annual bonus | | — | | — | | | 396,150 | | | 594,225 | | | — | | | — | | | — | | | — | |
| Eric Schmidt, Ph.D. | | | | | | | | | | | | | | | | |
| Restricted stock unit award | | 3/26/2021 | | — | | | — | | | — | | | 24,866 | | | — | | | — | | | 839,973 | |
| Stock option award | | 3/26/2021 | | — | | | — | | | — | | | — | | | 92,365 | | | 33.78 | | | 1,963,489 | |
| Annual bonus | | — | | — | | | 180,500 | | | 270,750 | | | — | | | — | | | — | | | — | |
| Rafael Amado, M.D. | | | | | | | | | | | | | | | | |
| Restricted stock unit award | | 3/26/2021 | | — | | | — | | | — | | | 24,866 | | | — | | | — | | | 839,973 | |
| Restricted stock unit award | | 5/18/2021 | | — | | | — | | | — | | | 200,534 | | | — | | | — | | | 5,999,977 | |
| Stock option award | | 3/26/2021 | | — | | | — | | | — | | | — | | | 92,365 | | | 33.78 | | | 1,963,489 | |
| Annual bonus | | — | | — | | | 228,000 | | | 342,000 | | | — | | | — | | | — | | | — | |
| Alison Moore, Ph.D. | | | | | | | | | | | | | | | | |
| Restricted stock unit award | | 3/26/2021 | | — | | | — | | | — | | | 24,866 | | | — | | | — | | | 839,973 | |
| Stock option award | | 3/26/2021 | | — | | | — | | | — | | | — | | | 92,365 | | | 33.78 | | | 1,963,489 | |
| Annual bonus | | — | | — | | | 180,500 | | | 270,750 | | | — | | | — | | | — | | | — | |
| Veer Bhavnagri | | | | | | | | | | | | | | | | |
| Restricted stock unit award | | 3/26/2021 | | — | | | — | | | — | | | 22,202 | | | — | | | — | | | 749,984 | |
| Stock option award | | 3/26/2021 | | — | | | — | | | — | | | — | | | 82,469 | | | 33.78 | | | 1,753,120 | |
| Annual bonus | | — | | — | | | 176,320 | | | 264,480 | | | — | | | — | | | — | | | — | |
(1) All stock options and restricted stock unit awards were granted under the EIP.
NARRATIVE TO SUMMARY COMPENSATION TABLE AND GRANTS OF PLAN-BASED AWARDS TABLE
Agreements with our Named Executive Officers
We have entered into employment letter agreements with each of our named executive officers.Named Executive Officers. These letter agreements generally provide forat-will employment and set forth the named executive officer’sNamed Executive Officer’s initial base salary, eligibility for employee benefits and recommended stock optionequity grant. In addition, each of our named executive officersNamed Executive Officers has executed a form of our standard confidential information and
invention assignment agreement. The key terms of the letter agreements with our
named executive officersNamed Executive Officers are described below. Any potential payments and benefits due upon a qualifying termination of employment or a change in control of the Company are further described below under
“- “—Potential Payments and Benefits upon Termination or Change in Control.”
David Chang, M.D., Ph.D. We entered into a letter agreement with Dr. Chang, our President and Chief Executive Officer, in June 2018 that governs the current terms of his employment with us. Pursuant to the agreement, Dr. Chang iswas entitled to an annual base salary of $525,000 which was increased to $600,000 for 2019. Dr. Changand was eligible to receive an annual target performance bonus of up to 45% of his. As described above, we have increased Dr. Chang's annual base salary for 2018 and is eligible to receive an annual target performance bonus of up to 55% of his base salary for 2019.over time. In addition, Dr. Chang was paid a $250,000 relocation bonus in 2018 and is eligible forwas paid an additional relocation bonus of $250,000 in 2019 in connection with his relocation to the San Francisco Bay Area. Dr. Chang was granted an initial stock option to purchase 1,955,625 shares of our common stock. Additionally, we entered into a vesting restriction agreement with Dr. Chang in April 2018, pursuant to which the 4,280,230 shares of common stock beneficially owned by Dr. Chang and issued in December 2017 became subject to vesting over a52-month period commencing in December 2017, subject to his continuous service through each vesting date.
Eric Schmidt, Ph.D. We entered into a letter agreement with Dr. Schmidt, our Chief Financial Officer, in June 2018 that governs the current terms of his employment with us. Pursuant to the agreement, Dr. Schmidt iswas entitled to an annual base salary of $375,000 which was increased to $425,000 for 2019. Dr. Schmidtand was eligible to receive an annual target performance bonus of up to 35%. As described above, we have increased Dr. Schmidt's annual base salary and annual target performance bonus over time. Dr. Schmidt was granted an initial stock option to purchase 1,464,750 shares of our common stock.
Rafael Amado, M.D. We entered into a letter agreement with Dr. Amado, our Chief Medical Officer and Executive Vice President of Research and Development, in July 2019 that governs the current terms of his employment with us. Pursuant to the agreement, Dr. Amado was entitled to an annual base salary for 2018of $500,000 and iswas eligible to receive an annual target performance bonus of up to 40% of his base salary. As described above, we have increased Dr. Amado's annual base salary for 2019.and annual target performance bonus over time. In addition, Dr. SchmidtAmado was paid a $75,000 sign-on advance and a $150,000 relocation bonus in 2019 in connection with his relocation to the San Francisco Bay Area. Dr. Amado was granted an initial stock option to purchase 1,464,750160,630 shares of our common stock. Dr. Amado was also granted an initial restricted stock unit award for 105,162 shares of our common stock, which settle on an annual basis over four years. In addition, Dr. Amado was awarded a new hire performance restricted stock unit award for 57,361 shares of our common stock, which settle in three equal installments, with (1) the first installment to vest upon initiation of a Phase 2 clinical trial of our first anti-CD19 product candidate, (2) the second installment to vest upon the filing of a biologics license application with the FDA for our first anti-CD19 product candidate, and (3) the third installment to vest upon FDA approval of our first anti-CD19 product candidate. Any unvested portion of the performance restricted stock unit will expire after seven years.
Alison Moore, Ph.D. We entered into a letter agreement with Dr. Moore, our Chief Technical Officer, in May 2018 that governs the current terms of her employment with us. Pursuant to the agreement, Dr. Moore iswas entitled27
to an annual base salary of $400,000 which was increased to $425,000 for 2019. Dr. Mooreand was eligible to receive an annual target performance bonus of up to 35% of her. As described above, we have increased Dr. Moore's annual base salary for 2018 and is eligible to receive an annual target performance bonus of up to 40% of her annual base salary for 2019.over time. In addition, Dr. Moore was paid a $100,000 relocation bonus in 2018 in connection with her relocation to the San Francisco Bay Area. Dr. Moore was granted an initial stock option to purchase 892,500 shares of our common stock.
Joshua Kazam
Veer Bhavnagri. We entered into a letter agreement with Mr. Bhavnagri, our General Counsel, in May 2018 that governs the current terms of his employment with us. Pursuant to the agreement, Mr. Bhavnagri was entitled to an annual base salary of $365,000 and was eligible to receive an annual target performance bonus of up to 35%. WeAs described above, we have increased Mr. Bhavnagri's annual base salary and annual target performance bonus over time. In addition, Mr. Bhavnagri was paid a $50,000 sign-on bonus and a $75,000 relocation bonus in 2018 in connection with his relocation to the San Francisco Bay Area. Mr. Bhavnagri was granted an initial stock option to purchase 367,500 shares of our common stock. Additionally, we entered into a vesting restriction agreement with Mr. KazamBhavnagri in April 2018, pursuant to which the 1,718,435262,500 shares of common stock beneficially owned by Mr. Kazam and issued in December 2017Bhavnagri became subject to vesting over a52-month period commencing in December 2017, subject to his continuous service through each vesting date. Each of the options granted to
Drs. Chang, Schmidt and Mooreour Named Executive Officers are subject to a four-year vesting schedule, with 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months, subject to each individual’s continued service through each vesting date.
Potential Payments
For additional information regarding our long-term compensation and Benefits upon Termination or Changeequity grants, please see "Executive Compensation--Executive Compensation Program and Compensation Decisions for the Named Executive Officers--Long-Term Incentive Compensation".
CEO Pay Ratio
Our compensation and benefits philosophy and the overall structure of
ControlRegardlessour compensation and benefit programs are broadly similar across the organization. We strive to provide pay, benefits, and services that are competitive to market and create incentives to attract and retain employees. Our compensation package includes market-competitive pay, broad-based stock grants, health care and 401(k) plan benefits, paid time off and family leave, among others. We also provide annual incentive bonus opportunities that are tied to both company performance as well as individual performance to foster a pay-for-performance culture.
Under rules adopted pursuant to the Dodd-Frank Act, we are required to calculate and disclose the total compensation paid to our median paid employee, as well as the ratio of the total compensation paid to the median employee as compared to the total compensation paid to our Chief Executive Officer (the “CEO Pay Ratio”). The paragraphs that follow describe our methodology and the resulting CEO Pay Ratio.
Measurement and Methodology
We identified the median employee using our employee population on December 31, 2021 (including all employees, whether employed on a full-time or part-time basis).
Under the relevant rules, we were required to identify the median employee by use of a “consistently applied compensation measure,” or CACM. We chose a CACM that closely approximates the total annual direct compensation of our employees. Specifically, we identified the median employee by looking at annual base pay, awarded annual cash incentive opportunity, and the grant date fair value for equity awards granted as of December 31, 2021 for all active employees as of that date. The value of our 401(k) plan and health and welfare benefits provided was excluded as all employees, including the Chief Executive Officer, are offered the same benefits. We did not perform adjustments to the compensation paid to part-time employees to calculate what they would have been paid on a full-time basis.
After applying our CACM methodology, we identified the median employee. Once the median employee was identified, we calculated the median employee’s annual target total direct compensation in accordance with the requirements of the Summary Compensation Table.
Pay Ratio
Our median employee compensation as calculated using Summary Compensation Table requirements was $307,290. Our Chief Executive Officer’s compensation as reported in the Summary Compensation Table was $10,161,454. Therefore, our CEO Pay Ratio is approximately 33:1.
This information is being provided for compliance purposes and is a reasonable estimate calculated in a manner consistent with SEC rules, based on our internal records and the methodology described above. The pay ratio reported by other companies may not be comparable to the pay ratio reported above, as other companies have different employee populations and compensation practices and may use different methodologies, exclusions, estimates and assumptions in calculating their own pay ratios. Neither the Compensation Committee nor management of the Company used the CEO Pay Ratio measure in making compensation decisions.
2021 OUTSTANDING EQUITY AWARDS AT FISCAL YEAR-END TABLE
The following table presents information concerning equity awards held by our Named Executive Officers as of December 31, 2021. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | Option Awards | | Stock Awards |
| Name | | Grant Date | | Number of Securities Underlying Unexercised Options (#)
Exercisable | | Number of Securities Underlying Unexercised Options (#)
Unexercisable | | Option Exercise Price
($) | | Option Expiration Date | | Number of Shares of Stock that have not Vested (#) | | Market Value of Shares of Stock that have not Vested ($) |
| David Chang, M.D., Ph.D.(1)(2)(5) | | 4/6/2018 | | — | | | — | | | — | | | — | | 246,938 | | | 3,684,315 | |
| 6/25/2018 | | — | | | — | | | 2.27 | | 6/25/2028 | | 162,969 | | | 2,431,497 | |
| 3/13/2019 | | 292,700 | | | — | | | 26.89 | | 3/13/2029 | | 86,500 | | | 1,290,580 | |
| 3/18/2020 | | 541,190 | | | — | | | 18.22 | | 3/18/2030 | | 12,544 | | | 187,156 | |
| 3/26/2021 | | 298,836 | | | — | | | 33.78 | | 3/26/2031 | | 80,452 | | | 1,200,344 | |
| | | | | | | | | | | | | |
| Eric Schmidt, Ph.D.(3)(6) | | 6/25/2018 | | — | | | — | | | 2.27 | | 6/25/2028 | | 183,094 | | | 2,731,762 | |
| 3/13/2019 | | 83,500 | | | — | | | 26.89 | | 3/13/2029 | | 24,700 | | | 368,524 | |
| 3/18/2020 | | 94,703 | | | — | | | 18.22 | | 3/18/2030 | | 35,644 | | | 531,808 | |
| 3/26/2021 | | 92,365 | | | — | | | 33.78 | | 3/26/2031 | | 24,866 | | | 371,001 | |
| | | | | | | | | | | | | |
| Rafael Amado, M.D.(4) | | 9/3/2019 | | 160,630 | | | — | | | 26.15 | | 9/3/2029 | | 109,942 | | | 1,640,335 | |
| 3/18/2020 | | 109,600 | | | — | | | 18.22 | | 3/18/2030 | | 23,100 | | | 344,652 | |
| 3/26/2021 | | 92,365 | | | — | | | 33.78 | | 3/26/2031 | | 24,866 | | | 371,001 | |
| 5/18/2021 | | — | | | — | | | — | | | — | | 200,534 | | | 2,991,967 | |
| | | | | | | | | | | | | |
| Alison Moore, Ph.D. | | 6/25/2018 | | 579,959 | | | — | | | 2.27 | | 6/25/2028 | | — | | | — | |
| 3/13/2019 | | 83,500 | | | — | | | 26.89 | | 3/13/2029 | | 18,525 | | | 276,393 | |
| 3/18/2020 | | 109,600 | | | — | | | 18.22 | | 3/18/2030 | | 23,100 | | | 344,652 | |
| 3/26/2021 | | 92,365 | | | — | | | 33.78 | | 3/26/2031 | | 24,866 | | | 371,001 | |
| | | | | | | | | | | | | |
| Veer Bhavnagri(1) | | 4/6/2018 | | — | | | — | | | — | | | — | | 15,144 | | | 225,952 | |
| 6/25/2018 | | 112,300 | | | — | | | 2.27 | | 6/25/2028 | | — | | | — | |
| 3/13/2019 | | 73,500 | | | — | | | 26.89 | | 3/13/2029 | | 21,700 | | | 323,764 | |
| 3/18/2020 | | 105,200 | | | — | | | 18.22 | | 3/18/2030 | | 22,125 | | | 330,105 | |
| 3/26/2021 | | 82,469 | | | — | | | 33.78 | | 3/26/2031 | | 22,202 | | | 331,254 | |
| | | | | | | | | | | | | |
(1) In connection with the issuance of the Company’s Series A and Series A-1 convertible preferred stock in April 2018, the Company’s founders, including Dr. Chang and Mr. Bhavnagri, agreed to modify their fully vested founders’ shares of common stock outstanding to include a forfeiture restriction that lapses based on their continued service to the Company. The number of shares of stock that have not vested is based on the founders’ shares that were beneficially owned by Dr. Chang and Mr. Bhavnagri as of December 31, 2021. The shares are subject to vesting over a 52-month period commencing in December 2017, subject to continuous service through each vesting date.
(2) On July 9, 2018, Dr. Chang elected to early exercise in full his stock option granted on June 25, 2018. We have a right to repurchase any unvested shares subject to such award if Dr. Chang ceases to provide services to us prior to the date on which all shares subject to the award have vested in accordance with the applicable vesting schedule. The June 25, 2018 option award is subject to a four-year vesting schedule, with 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months. The vesting commencement date for such award was April 6, 2018.
(3) On June 26, 2018, Dr. Schmidt elected to early exercise in full his stock option granted on June 25, 2018. We have a right to repurchase any unvested shares subject to such award if Dr. Schmidt ceases to provide services to us prior to the date on which all shares subject to the award have vested in accordance with the applicable vesting schedule. The June 25, 2018 option award is subject to a four-year vesting schedule, with 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months. The vesting commencement date for such award was June 18, 2018.
(4) A total of 57,361 of the stock awards granted on September 3, 2019 are performance based restricted stock units which vest upon achievement of specific performance criteria.
(5) On May 24, 2021, Dr. Chang elected to early exercise 14,897 of his stock option granted on March 18, 2020. We have a right to repurchase any unvested shares subject to such award if Dr. Chang ceases to provide services to us prior to the date on which all shares subject to the award have vested in accordance with the applicable vesting schedule. The March 18, 2020 option award is subject to a four-year vesting schedule, with 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months. The vesting commencement date for such award was March 18, 2020.
(6) On May 24, 2021, Dr. Schmidt elected to early exercise 14,897 of his stock option granted on March 18, 2020. We have a right to repurchase any unvested shares subject to such award if Dr. Schmidt ceases to provide services to us prior to the date on which all shares subject to the award have vested in accordance with the applicable vesting schedule. The March 18, 2020 option award is subject to a four-year vesting schedule, with 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months. The vesting commencement date for such award was March 18, 2020.
2021 OPTION EXERCISES AND STOCK VESTED TABLE
The following table provides information on stock options exercised, including the number of shares of our common stock acquired upon exercise and the value realized, determined as described below, for our Named Executive Officers in the year ended December 31, 2021:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Option Awards | | Stock Awards |
| Name | | Number of Shares Acquired on Exercise (#) | | Value Realized on Exercise ($)(1) | | Number of Shares Acquired on Vesting (#) | | Value Realized on Vesting
($) |
| David Chang, M.D., Ph.D.(2) | | 27,410 | | | 190,774 | | | 987,756 | | | 25,389,434 | |
| Eric Schmidt, Ph.D. | | 14,897 | | | 103,683 | | | 386,238 | | | 10,216,429 | |
| Rafael Amado, M.D. | | — | | | — | | | 33,991 | | | 971,975 | |
| Alison Moore, Ph.D. | | — | | | — | | | 16,963 | | | 652,736 | |
| Veer Bhavnagri(2) | | 50,000 | | | 1,339,753 | | | 78,802 | | | 2,258,378 | |
| | | | | |
| (1) | The value realized on exercise is based on the difference between the closing market price of our common stock on the date of exercise and the exercise price of the applicable options, and does not represent actual amounts received by the Named Executive Officers as a result of the option exercises. |
| (2) | In connection with the issuance of the Company’s Series A and Series A-1 convertible preferred stock in April 2018, the Company’s founders, including Dr. Chang and Mr. Bhavnagri, agreed to modify their fully vested founders’ shares of common stock outstanding to include a forfeiture restriction that lapses based on their continued service to the Company. The shares are subject to vesting over a 52-month period commencing in December 2017, subject to continuous service through each vesting date. The shares vested in this table include a portion of the founders' shares that vested during 2021. |
2021 DIRECTOR COMPENSATION TABLE
The following table shows for the fiscal year ended December 31, 2021 certain information with respect to the compensation of all non-employee directors of the Company that served during 2021: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name | | Fees earned ($) | | Stock awards ($)(1) | | Option awards ($)(2) | | All other Compensation ($)(3) | | Total ($) |
| Elizabeth Barrett | | 18,750 | | | — | | 844,582 | | — | | | 863,332 |
| Arie Belldegrun, M.D. | | — | | | 1,811,790 | | 4,235,069 | | 726,910 | | | 6,773,769 |
| David Bonderman | | — | | | — | | — | | — | | | — |
| John DeYoung | | — | | | — | | — | | — | | | — |
| Franz Humer, Ph.D. | | 163,125 | | | 424,988 | | — | | — | | | 588,113 |
| Joshua Kazam | | 54,000 | | | 424,988 | | — | | 603,160 | | | 1,082,148 |
| Deborah Messemer | | 66,875 | | | — | | 424,988 | | — | | | 491,863 |
| Vicki Sato, Ph.D. | | 20,167 | | | 424,996 | | 422,284 | | — | | | 867,447 |
| Todd Sisitsky | | — | | | — | | — | | — | | | — |
| Owen Witte, M.D | | 50,000 | | | — | | 424,988 | | 27,233 | | | 502,221 |
(1) The amounts reported in this column represent the aggregate grant date fair value of the restricted stock unit awards granted during the year ended December 31, 2021, as computed in accordance with Financial Accounting Standard Board Accounting Standards Codification Topic 718 for stock-based compensation transactions (ASC Topic 718) excluding the impact of estimated forfeitures related to service-based vesting conditions, which value is based on the closing market price of our common stock on the date of grant. As of December 31, 2021, the aggregate number of restricted stock units outstanding held by our non-employee directors were: Dr. Belldegrun: 111,302; Dr. Humer: 16,498; Mr. Kazam: 16,498; and Dr. Sato: 19,362.
(2) The dollar amounts in this column represent the aggregate grant date fair value of stock option awards granted in 2021. These amounts have been computed in accordance with FASB ASC Topic 718, using the Black-Scholes option pricing model. For a discussion of valuation assumptions, see Note 10 “Stock-based Compensation” to our financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2021. These amounts do not reflect the actual economic value that will be realized by the directors upon the vesting of the stock options, the exercise of the stock options, or the sale of the common stock underlying such stock options. As of December 31, 2021, the aggregate number of shares outstanding under all option awards held by our non-employee directors were: Ms. Barrett: 63,385; Dr. Belldegrun: 394,356; Dr. Humer: 183,750; Ms. Messemer: 280,680; Dr. Sato: 34,692; and Dr. Witte: 54,040.
(3) For Dr. Belldegrun, amount shown represents $463,000 in consulting fees paid to Bellco Capital LLC (“Bellco”) in 2021 and an executive officer’s service terminates, each executive officerannual performance award of $263,910 paid to Bellco in 2022 for services rendered in 2021. Bellco is entitledowned by Dr. Belldegrun, as co-trustee of the Belldegrun Family Trust, and Dr. Rebecka Belldegrun, as co-trustee of the Belldegrun Family Trust. For Mr. Kazam, amount shown represents consulting fees paid to receive amountsTwo River, LLC (“Two River”) in 2021. Mr. Kazam provides consulting services to us through Two River and is a venture partner of Two River. For more information, see description of our consulting arrangement with Bellco and Two River below under “Transactions With Related Persons—Consulting Arrangements”. For Dr. Witte, amount shown represents $27,233 in fees earned during his or her termin connection with Dr. Witte's services on our scientific advisory board.
We have reimbursed and will continue to reimburse all of service, including unpaid salaryour non-employee directors for their travel, lodging and unused paid-time off, as applicable. In addition,other reasonable expenses incurred in attending meetings of our Board hasof Directors and committees of our Board of Directors, and will pay for the travel, lodging and other reasonable expenses incurred by our employee directors to attend meetings of our Board of Directors and, as applicable, committees of our Board of Directors.
Executive Chair
Dr. Belldegrun serves as our Executive Chair and also provides consulting services through Bellco. Dr. Belldegrun contributes comprehensive, in-depth knowledge of our business, cell therapy generally and the global life sciences industry, as well as valuable insights on leadership and strategy. Our Board believes that his advice and guidance to our senior leadership team and Board is essential to advancing our business.
Specifically, Dr. Belldegrun is actively involved as both Executive Chair of the Board and strategic consultant, including through the following activities:
•Presiding over all Board meetings;
•Attending weekly senior leadership team meetings and off-site strategy meetings;
•Reviewing and approving Board agenda items;
•Helping to recruit and assess potential directors;
•Overseeing Board and director evaluations;
•Reviewing Chief Executive Officer performance;
•Providing strategic advice to the Chief Executive Officer, as well as the rest of the senior leadership team and our Board, including on operational, financial and strategic matters;
•Assisting with efforts to recruit and retain key executive talent;
•Assisting management in growing Allogene and advancing clinical trials;
•Communicating efforts with investors;
•Contributing key scientific and medical knowledge; and
•Assisting with key relationships, including collaborators, licensors and clinical trial sites.
Given Dr. Belldegrun's critical importance and deep involvement in Allogene, the Compensation Committee recommended and our Board approved an equity grant in March 2021 that was approximately two-thirds the value of the grant to our CEO. Any compensation matters and decisions relating to Dr. Belldegrun and Bellco are reviewed outside of Dr. Belldegrun's presence by both the Compensation Committee and the Board, and Dr. Belldegrun does not participate in the deliberations or determination of his own compensation.
Non-Employee Director Compensation Policy
Our non-employee director compensation policy includes:
•an annual cash retainer of $40,000;
•an additional annual cash retainer of $12,500, $7,500 and $5,000 for service as a Changemember of the Audit Committee, Compensation Committee and the Nominating and Corporate Governance Committee, respectively;
•an additional annual cash retainer of $100,000, $25,000, $15,000 and $10,000 for service as chair of the International and Business Development Oversight Committee, Audit Committee, Compensation Committee and the Nominating and Corporate Governance Committee, respectively (in lieu of the committee member retainer above);
•an additional cash payment of $3,500 per meeting for each other member of the International and Business Development Oversight Committee;
•an initial option grant, vesting in Control Plan described below.36 equal monthly installments, and/or an initial restricted stock unit award, vesting annually over a three-year period from the date of grant, having an aggregate grant date value of $850,000, with the director designating the proportionate share between the initial option grant and initial restricted stock unit award prior to or on the date of grant; and
•an annual option grant, vesting in 12 equal monthly installments, and/or an annual restricted stock unit award, vesting on the one-year anniversary of the date of grant, having an aggregate grant date value of $425,000, with the director designating the proportionate share between the annual option grant and annual restricted stock unit award prior to or on the date of grant. The annual grants will be made on the date of each of our annual meetings of stockholders.
Each of the equity awards described above will vest and become exercisable subject to the non-employee director’s continuous service with us through each applicable vesting date, provided that each option and restricted stock unit award will vest in full upon a change of control of the Company, as defined under our named executive officers holdsEIP. The stock options and RSUrestricted stock unit awards will be granted under our EIP.
In consultation with Compensia, the Compensation Committee and specifically Mr. Bonderman and Mr. DeYoung reviewed the non-employee director compensation policy and concluded that no changes were warranted in 2021. Neither Mr. Bonderman nor Mr. DeYoung accept any compensation for serving on our Board of Directors.
SECURITIES AUTHORIZED FOR ISSUANCE UNDER EQUITY COMPENSATION PLANS
The following table sets forth the aggregate information of our equity compensation plans in effect as of December 31, 2021.
Equity Compensation Plan Information
| | | | | | | | | | | | | | | | | | | | |
| Plan Category | | Number of securities to be issued upon exercise of outstanding options, warrants and rights
(a) | | Weighted-average exercise price of outstanding options, warrants and rights
(b)($) | | Number of securities remaining available for issuance under equity compensation plans (excluding securities reflected in column (a))
(c) |
| Equity compensation plans approved by security holders | | — | | | — | | | — | |
| Amended and Restated 2018 Equity Incentive Plan (Prior Plan)(1) | | 2,454,684 | | | 4.78 | | | — | |
| Amended and Restated 2018 Equity Incentive Plan(2) | | 12,045,591 | | | 21.10 | | | 15,801,927 | |
| 2018 Employee Stock Purchase Plan(3) | | — | | | — | | | 4,551,912 | |
| Equity compensation plans not approved by security holders | | — | | | — | | | — | |
| Total | | 14,500,275 | | | 25.88 | | | 20,353,839 | |
(1) As of December 31, 2021, under our Amended and Restated 2018 Equity Incentive Plan (the “Prior Plan”), the number of outstanding awards under column (a) includes 2,454,684 shares which are issuable upon the exercise of outstanding options (including options that are immediately exercisable) at a weighted-average exercise price of $4.78.
(2) The EIP became effective on September 26, 2018, which replaced the Prior Plan. Initially, the aggregate number of shares of our equitycommon stock that may be issued under the EIP is 20,432,250 shares. Additionally, on January 1 of each year, commencing on January 1, 2019 and ending on January 1, 2028, the number of shares authorized for issuance under the 2018 Plan is automatically increased by a number equal to: (a) 5% of the total number of shares of capital stock outstanding on December 31 of the preceding calendar year; or (b) such lesser number of shares of common stock as is determined by our Board or the Compensation Committee for the applicable year. All shares of our common stock reserved and available under the EIP shall constitute the maximum aggregate number of shares of common stock that may be issued through incentive plans that werestock options. As of December 31, 2021, the number of outstanding awards under column (a) includes: (1) 10,239,167 shares issuable upon the exercise of outstanding options at a weighted-average exercise price of approximately $21.10 and (2) 4,261,108 shares issuable upon the vesting of outstanding restricted stock units. The weighted-average exercise price shown in column (b) is for the outstanding options only. As of January 1, 2022, the number of shares issuable under the EIP increased to 46,874,618 shares, of which 22,933,080 shares remained available for issuance under the EIP.
(3) Our 2018 Employee Stock Purchase Plan (the “ESPP”) became effective on September 26, 2018. The ESPP authorizes the issuance of 1,160,000 shares of our common stock pursuant to purchase rights granted subject to our formemployees or to employees of stock optionany of our designated affiliates. On January 1 of each year, commencing on January 1, 2019 and RSU agreements. A descriptionending on January 1, 2028, the number of shares authorized for issuance under the 2018 ESPP is automatically increased by a number equal to the lesser of: (a) 1% of the termination and changetotal number of control provisions inshares of capital stock outstanding on December 31 of the preceding calendar year; (b) 2,320,000 shares; or (c) such equity incentive plans and equity awards granted thereunderlesser number of shares of Common Stock as is provided below under “– Equity Benefit Plans” anddetermined by our Board or the specific vesting termsCompensation Committee for the applicable year. The ESPP is intended to qualify as an “employee stock purchase plan” within the meaning of each namedSection 423 of the Code.
POTENTIAL PAYMENTS UPON TERMINATION OR CHANGE IN CONTROL
Our executive
officer’s equity awards are described below under “– Outstanding Equity Awards at FiscalYear-End.Change in Control and Severance Benefit Plan
Our current executive officers, including our Named Executive Officers, are entitled to certain severance and change of control payments and benefits pursuant to our change in control and severance benefit plan (the “Change in Control Plan”). The Change in Control Plan provides for, in the event of an involuntary termination of employment without “cause” or a resignation with “good reason,” and subject to our receipt of an effective waiver and release of claims from the executive, a combination of (1) cash severance for the severance period and (2) the payment or reimbursement of premiums or continuedfor continuation of coverage under group health plans for the severance period. The severance period is 24 months in the case of our Chief Executive Officer, and 12 months in the case of our Chief Financial Officer and Chief Technical Officer.
other Named Executive Officers.
In the event that the involuntary termination of employment occurs within the period commencing three months before and ending 12 months after a change in control of the Company, then the participants in the Change in Control Plan are entitled to the same benefits described above, but the severance period is 18 months for our Chief Financial Officer,
Chief Medical Officer and
Executive Vice President of Research and Development, Chief Technical
Officer.Officer and General Counsel. In addition, our Chief Executive Officer would be entitled to 200% of his annual target bonus and our
Chief Financial Officer and Chief Technical Officerother Named Executive Officers would be entitled to 150% of
his or hereach Named Executive Officer's annual target bonus, and each of our
executive officersNamed Executive Officers would be entitled to accelerated vesting of outstanding equity compensation awards.
Under the Change in Control Plan, the term “cause” generally means (i) the employee’s commission of any crime involving fraud, dishonesty or moral turpitude; (ii) the employee’s attempted commission of or participation in a fraud or act of dishonesty against us
that results in (or might have reasonably resulted in) material harm to our business; (iii) the employee’s intentional, material violation of any contract or agreement between us and the employee or any statutory duty that the employee owes to us; or (iv) the employee’s conduct that constitutes gross insubordination, incompetence or habitual neglect of duties and that results in (or might have reasonably resulted in) material harm to our business. The term “change in control” generally means (1) the acquisition by
28
any person or company of more than 50% of the combined voting power of our then outstanding stock, (2) a merger, consolidation or similar transaction in which our stockholders immediately before the transaction do not own, directly or indirectly, more than 50% of the combined voting power of the surviving entity (or the parent of the surviving entity) in substantially the same proportions as their ownership immediately prior to such transaction, (3) a sale, lease, exclusive license or other disposition of all or substantially all of our assets other than to an entity more than 50% of the combined voting power of which is owned by our stockholders in substantially the same proportions as their ownership of our outstanding voting securities immediately prior to such transaction, or (4) a complete dissolution or liquidation of the company.
The term “good reason” generally means (i) a material reduction of such employee’s annual base salary, which is a reduction of at least 10% of such employee’s base salary (unless pursuant to a salary reduction program applicable generally to the Company’s similarly situated employees); (ii) a material reduction in such employee’s authority, duties or responsibilities; (iii) a relocation of such employee’s principal place of employment with the Company (or successor to the Company, if applicable) to a place that increases such employee’s
one-way commute by more than 50 miles as compared to such employee’s then-current principal place of employment immediately prior to such relocation (excluding regular travel in the ordinary course of business).
2018 Outstanding Equity Awards at FiscalYear-End Table
The following table
presents information concerning equity awards held by our named executive officers as of December 31, 2018. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Option Awards | | | Stock Awards | |
| Name | | Grant Date | | | Number of
Securities
Underlying
Unexercised
Options (#)
Exercisable | | | Number of
Securities
Underlying
Unexercised
Options (#)
Unexercisable | | | Option
Exercise
Price ($) | | | Option
Expiration
Date | | | Number of
Shares or
Units of
Stock That
Have Not
Vested (#) | | | Market
Value
of Shares
or Units
of Stock
That Have
Not Vested
($) | |
David Chang, M.D., Ph.D. | | (1) | | | — | | | | — | | | | — | | | | — | | | | — | | | | 2,075,952 | | | | 55,905,387 | |
| | (2) | | | — | | | | — | | | | — | | | | — | | | | — | | | | 1,955,625 | | | | 52,664,981 | |
Joshua Kazam | | (1) | | | — | | | | — | | | | — | | | | — | | | | — | | | | 1,028,951 | | | | 27,709,650 | |
Eric Schmidt, Ph.D. | | (3) | | | — | | | | — | | | | — | | | | — | | | | — | | | | 1,464,750 | | | | 39,445,718 | |
Alison Moore, Ph.D. | | (4) | | | 6/25/2018 | | | | 892,500 | | | | — | | | | 2.26 | | | | 6/25/2028 | | | | — | | | | — | |
(1) | In connection with the issuance of the Company’s Series A and SeriesA-1 convertible preferred stock in April 2018, the Company’s founders, including Dr. Chang and Mr. Kazam, agreed to modify their fully vested founders’ shares of common stock outstanding to include a forfeiture restriction that lapses based on their continued service to the Company. The number of shares of stock that have not vested is based on the founders’ shares that were beneficially owned by Dr. Chang and Mr. Kazam as of December 31, 2018. The shares are subject to vesting over a52-month period commencing in December 2017, subject to continuous service through each vesting date.
|
(2) | On July 9, 2018, Dr. Chang elected to early exercise in full his stock option granted on June 25, 2018. We have a right to repurchase any unvested shares subject to such award if Dr. Chang ceases to provide services to us prior to the date on which all shares subject to the award have vested in accordance with the applicable vesting schedule. The June 25, 2018 option award is subject to a four-year vesting schedule, with 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months. The vesting commencement date for such award is April 6, 2018.
|
(3) | On June 26, 2018, Dr. Schmidt elected to early exercise in full his stock option granted on June 25, 2018. We have a right to repurchase any unvested shares subject to such award if Dr. Schmidt ceases to provide services to us prior to the date on which all shares subject to the award have vested in accordance with the applicable vesting schedule. The June 25, 2018 option award is subject to a four-year vesting schedule, with
|
29
| 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months. The vesting commencement date for such award is June 18, 2018. |
(4) | Stock option is subject to a four-year vesting schedule, with 25% vesting one year after the vesting commencement date and the balance vesting monthly over the remaining 36 months, subject to continued service through each vesting date. The vesting commencement date is June 1, 2018.
|
Policy Against Hedging and Speculative Trading and Pledging our Common Stock
Our insider trading policy prohibits our employees from engaging in “hedging” or other inherently speculative transactions with respect to our common stock or borrowing against our common stock.
Stock Ownership Guidelines Policy
In March 2019, our Compensation Committee recommended and our Board adopted a Stock Ownership Guidelines Policy. The purposeprovides an estimate of the Guidelines is to encourage ownership of our common stock, promote the alignment of the long-term interests of our Chief Executive Officer, Chief Financial Officer, Chief Technical Officerpotential payments and directors with the long-term interests of the Company’s stockholders, and further promote our commitment to sound corporate governance. Under the Guidelines, the target common stock ownership level for our President and Chief Executive Officer is three times (3x) his base annual salary, the target stock ownership level for ournon-executive directors is three times (3x) their base annual cash retainer and the target stock ownership level for our other executive officers is one times (1x) their base annual salary. Under these Guidelines, the compliance deadline for all of our current executive officers and directors is December 2023, although we expect that the target stock ownership levels likely will be achieved much sooner than that.
Health, Welfare and Retirement Benefits; Perquisites
Our executive officers, during their employment with us, are eligible to participate in our employee benefit plans, including our medical, dental, group term life, disability and accidental death and dismemberment insurance plans, in each case on the same basis as all of our other employees. In addition, we provide a Section 401(k) plan to our employees, including our executive officers, as discussed in the section below entitled “— 401(k) Plan.”
We generally do not provide perquisites or personal benefits to our executive officers. We do, however, pay the premiums for medical, dental, group term life, disability and accidental death and dismemberment insurance for all of our employees. Our Board may elect to adopt qualified or nonqualified benefit plans in the future if it determines that doing so is in our best interests.
401(k) Plan
We maintain a defined contribution employee retirement plan (the “401(k) Plan”), for our employees. Our executive officers are eligible to participate in the 401(k) Plan on the same basis as our other employees. The 401(k) Plan is intended to qualify as atax-qualified plan under Section 401(a) of the Code. The 401(k) Plan provides that each participant may contribute up to the lesser of 100% of his or her compensation or the statutory limit, which was $18,500 for calendar year 2018. Participants that are 50 years or older can also make“catch-up” contributions, which in calendar year 2018 may have been up to an additional $6,000 above the statutory limit. We currently make matching contributions into the 401(k) Plan on behalf of participants. We match 100% of eligible contributions up to the first 3%, with an additional match of 50% on the next 3% (maximum of 4.5%). Participant contributions are held and invested, pursuant to the participant’s instructions, byChange in Control Plan, which could occur upon termination of the plan’s trustee. Our named executive officers did not contribute to the 401(k) Plan during 2018 and therefore did not receive any matching contributions during 2018.
30
Equity Benefit Plans
Amended and Restated 2018 Equity Incentive Plan
Our Board adoptedemployment of our 2018 Plan in September 2018 and our stockholders approved our 2018 Plan in October 2018. Our 2018 Plan became effective on October 2, 2018Named Executive Officers, including in connection with our initial public offering. Our 2018 Plan is a successor to and continuationchange of our Prior Plan. No further grants will be made undercontrol of the Prior Plan. As of
Company, assuming a triggering event occurred on December 31, 2018, there were 8,176,125 shares remaining available for2021: | | | | | | | | | | | | | | | | | | | | |
| Name | | Benefit | | Termination without Cause or Resignation for Good Reason Not in Connection with a Change in Control
($) | | Termination without Cause or Resignation for Good Reason in Connection with a Change in Control
($) |
| David Chang, M.D., Ph.D. | | Lump Sum Cash Severance Payment | | 1,390,000 | | | 1,390,000 | |
| | Lump Sum Target Bonus Payment | | — | | | 834,000 | |
| | Health Insurance Premiums | | 35,886 | | | 35,886 | |
| | Vesting Acceleration(1) | | — | | | 4,552,482 | |
| | Benefit Total | | 1,425,886 | | | 6,812,368 | |
| Eric Schmidt, Ph.D. | | Lump Sum Cash Severance Payment | | 475,000 | | | 712,500 | |
| | Lump Sum Target Bonus Payment | | — | | | 285,000 | |
| | Health Insurance Premiums | | 29,787 | | | 44,680 | |
| | Vesting Acceleration(1) | | — | | | 3,400,316 | |
| | Benefit Total | | 504,787 | | | 4,442,496 | |
| Rafael Amado, M.D. | | Lump Sum Cash Severance Payment | | 600,000 | | | 900,000 | |
| | Lump Sum Target Bonus Payment | | — | | | 360,000 | |
| | Health Insurance Premiums | | 18,322 | | | 27,483 | |
| | Vesting Acceleration(1) | | — | | | 5,347,955 | |
| | Benefit Total | | 618,322 | | | 6,635,438 | |
| Alison Moore, Ph.D. | | Lump Sum Cash Severance Payment | | 475,000 | | | 712,500 | |
| | Lump Sum Target Bonus Payment | | — | | | 285,000 | |
| | Health Insurance Premiums | | 26,571 | | | 39,856 | |
| | Vesting Acceleration(1) | | — | | | 2,403,330 | |
| | Benefit Total | | 501,571 | | | 3,440,686 | |
| Veer Bhavnagri | | Lump Sum Cash Severance Payment | | 464,000 | | | 696,000 | |
| | Lump Sum Target Bonus Payment | | — | | | 278,400 | |
| | Health Insurance Premiums | | 18,322 | | | 27,483 | |
| | Vesting Acceleration(1) | | — | | | 1,372,554 | |
| | Benefit Total | | 482,322 | | | 2,374,437 | |
(1) The value of the future grantaccelerated vesting of stock awards under our 2018 Plan. As of December 31, 2018, there werethe outstanding stock options covering a total of 7,235,545 shares of our common stock that were granted under our 2018 Plan.Stock Awards.Our 2018 Plan provides for the grant of incentive stock options (“ISOs”) within the meaning of Section 422 of the Code, to employees, including employees of any parent or subsidiary, and for the grant of nonstatutory stock options (“NSOs”) stock appreciation rights, restricted stock awards, restricted stock unit awards performance stock awards, performance cash awards and other forms of stock awards to employees, directors and consultants, including employees and consultants of our affiliates. We have granted stock options and RSU awards under the 2018 Plan.
Authorized Shares.The maximum number of shares of our common stock that may be issued under our 2018 Plan is 20,432,250 shares, which is the sum of (1) 8,223,097 new shares, plus (2) the number of shares (not to exceed 12,209,153 shares) (i) that remained available for the issuance of awards under our Prior Plan at the time our 2018 Plan became effective, and (ii) any shares subject to outstanding stock options or other stock awards that were granted under our Prior Plan that terminate or expire prior to exercise or settlement; are forfeited because of the failure to vest; or are reacquired or withheld (or not issued) to satisfy a tax withholding obligation or the purchase or exercise price. In addition, the number of shares of our common stock reserved for issuance under our 2018 Plan will automatically increase on January 1 of each calendar year, starting on January 1, 2019 through January 1, 2028, in an amount equal to 5% of the total number of shares of our capital stock outstanding on the last day of the calendar month before the date of each automatic increase, or a lesser number of shares determined by our Board. The maximum number of shares of our common stock that may be issued on the exercise of ISOs under our 2018 Plan is 40,864,500.
Shares subject to stock awards granted under our 2018 Plan that expire or terminate without being exercised in full or that are paid out in cash rather than in shares do not reduce the number of shares available for issuance under our 2018 Plan. If any shares of common stock issued pursuant to a stock award are forfeited back to or repurchased or reacquired by us for any reason, the shares that are forfeited or repurchased or reacquired will revert to and again become available for issuance under the 2018 Plan. Any shares reacquired in satisfaction of tax withholding obligations or as consideration for the exercise or purchase price of a stock award will again become available for issuance under the 2018 Plan.
Plan Administration.Our Board, or a duly authorized committee of our Board, has the authority to administer our 2018 Plan and is referred to as the “plan administrator” herein. Our Board may also delegate to one or more of our officers the authority to (1) designate employees (other than officers) to receive specified stock awards and (2) determine the number of shares subject to such stock awards. Under our 2018 Plan, our Board has the authority to determine award recipients, grant dates, the numbers and types of stock awards to be granted, the applicable fair market value, and the provisions of each stock award, including the period of exercisability and the vesting schedule applicable to a stock award.
Under the 2018 Plan, the Board also generally has the authority to effect, with the consent of any adversely affected participant, (A) the reduction of the exercise, purchase, or strike price of any outstanding award; (B) the cancellation of any outstanding award and the grant in substitution therefore of other awards, cash, or other consideration; or (C) any other action that is treated as a repricing under generally accepted accounting principles.
31
Stock Options.ISOs and NSOs are granted under stock option agreements adopted by the plan administrator. The plan administrator determines the exercise price for stock options, within the terms and conditions of the 2018 Plan, provided that the exercise price of a stock option generally cannot be less than 100% of the fair market value of our common stock on the date of grant. Options granted under the 2018 Plan vest at the rate specified in the stock option agreement as determined by the plan administrator.
The plan administrator determines the term of stock options granted under the 2018 Plan, up to a maximum of 10 years. Unless the terms of an optionholder’s stock option agreement provide otherwise, if an optionholder’s service relationship with us or any of our affiliates ceases for any reason other than disability, death, or cause, the optionholder may generally exercise any vested options for a period of three months following the cessation of service. This period may be extended in the event that exercise of the option is prohibited by applicable securities laws or our insider trading policy. If an optionholder’s service relationship with us or any of our affiliates ceases due to death, or an optionholder dies within a certain period following cessation of service, the optionholder or a beneficiary may generally exercise any vested options for a period of 18 months following the date of death. If an optionholder’s service relationship with us or any of our affiliates ceases due to disability, the optionholder may generally exercise any vested options for a period of 12 months following the cessation of service. In the event of a termination for cause, options generally terminate upon the termination date. In no event may an option be exercised beyond the expiration of its term.
Acceptable consideration for the purchase of common stock issued upon the exercise of a stock option will be determined by the plan administrator and may include (1) cash, check, bank draft or money order, (2) a broker- assisted cashless exercise, (3) the tender of shares of our common stock previously owned by the optionholder, (4) a net exercise of the option if it is an NSO, or (5) other legal consideration approved by the plan administrator.
Unless the plan administrator provides otherwise, options generally are not transferable except by will or the laws of descent and distribution. Subject to approval of the plan administrator or a duly authorized officer in each case, (i) an option may be transferred pursuant to a domestic relations order, official marital settlement agreement, or other divorce or separation instrument and (ii) an optionholder may designate a beneficiary who may exercise the option following the optionholder’s death.
Tax Limitations on ISOs.The aggregate fair market value, determined at the time of grant, of our common stock with respect to ISOs that are exercisable for the first time by an award holder during any calendar year under all of our stock plans may not exceed $100,000. Options or portions thereof that exceed such limit will generally be treated as NSOs. No ISO may be granted to any person who, at the time of the grant, owns or is deemed to own stock possessing more than 10% of our total combined voting power or that of any of our affiliates unless (1) the option exercise price is at least 110% of the fair market value of the stock subject to the option on the date of grant, and (2) the term of the ISO does not exceed five years from the date of grant.
Restricted Stock Unit Awards.Restricted stock unit awards are granted under restricted stock unit award agreements adopted by the plan administrator. Restricted stock unit awards may be granted in consideration for any form of legal consideration that may be acceptable to our Board and permissible under applicable law. A restricted stock unit award may be settled by cash, delivery of stock, a combination of cash and stock as deemed appropriate by the plan administrator, or in any other form of consideration set forth in the restricted stock unit award agreement. Additionally, dividend equivalents may be credited in respect of shares covered by a restricted stock unit award. Except as otherwise provided in the applicable award agreement, restricted stock unit awards that have not vested will be forfeited once the participant’s continuous service ends for any reason.
Restricted Stock Awards.Restricted stock awards are granted under restricted stock award agreements adopted by the plan administrator. A restricted stock award may be awarded in consideration for cash, check, bank draft or money order, past or future services to us, or any other form of legal consideration that may be acceptable to our Board and permissible under applicable law. The plan administrator determines the terms and conditions of
32
restricted stock awards, including vesting and forfeiture terms. If a participant’s service relationship with us ends for any reason, we may receive any or all of the shares of common stock held by the participant that have not vested as of the date the participant terminates service with us through a forfeiture condition or a repurchase right.
Stock Appreciation Rights.Stock appreciation rights are granted under stock appreciation right agreements adopted by the plan administrator. The plan administrator determines the purchase price or strike price for a stock appreciation right, which generally cannot be less than 100% of the fair market value of our common stock on the date of grant. A stock appreciation right granted under the 2018 Plan vests at the rate specified in the stock appreciation right agreement as determined by the plan administrator.
The plan administrator determines the term of stock appreciation rights granted under the 2018 Plan, up to a maximum of 10 years. If a participant’s service relationship with us or any of our affiliates ceases for any reason other than cause, disability, or death, the participant may generally exercise any vested stock appreciation right for a period of three months following the cessation of service. This period may be further extended in the event that exercise of the stock appreciation right following such a termination of service is prohibited by applicable securities laws or our insider trading policy. If a participant’s service relationship with us, or any of our affiliates, ceases due to disability or death, or a participant dies within a certain period following cessation of service, the participant or a beneficiary may generally exercise any vested stock appreciation right for a period of 12 months in the event of disability and 18 months in the event of death. In the event of a termination for cause, stock appreciation rights generally terminate immediately upon the occurrence of the event giving rise to the termination of the individual for cause. In no event may a stock appreciation right be exercised beyond the expiration of its term.
Performance Awards.The 2018 Plan permits the grant of performance-based stock and cash awards. Our compensation committee may structure awards so that the stock or cash will be issued or paid only following the achievement of certainpre-established performance goals during a designated performance period.
The performance goals that may be selected include one or more of the following: (i) sales; (ii) revenues; (iii) assets; (iv) expenses; (v) market penetration or expansion; (vi) earnings from operations; (vii) earnings before or after deduction for all or any portion of interest, taxes, depreciation, amortization, incentives, service fees or extraordinary or special items, whether or not on a continuing operations or an aggregate or per share basis; (viii) net income or net income per common share (basic or diluted); (ix) return on equity, investment, capital or assets; (x) one or more operating ratios; (xi) borrowing levels, leverage ratios or credit rating; (xii) market share; (xiii) capital expenditures; (xiv) cash flow, free cash flow, cash flow return on investment, or net cash provided by operations; (xv) stock price, dividends or total stockholder return; (xvi) development of new technologies or products; (xvii) sales of particular products or services; (xviii) economic value created or added; (xix) operating margin or profit margin; (xx) customer acquisition or retention; (xxi) raising or refinancing of capital; (xxii) successful hiring of key individuals; (xxiii) resolution of significant litigation; (xxiv) acquisitions and divestitures (in whole or in part); (xxv) joint ventures and strategic alliances; (xxvi) spin-offs,split-ups and the like; (xxvii) reorganizations; (xxviii) recapitalizations, restructurings, financings (issuance of debt or equity) or refinancings; (xxix) or strategic business criteria, consisting of one or more objectives based on the following goals: achievement of timely development, design management or enrollment, meeting specifiedclosing market penetration or value added, payor acceptance, patient adherence, peer reviewed publications, issuance of new patents, establishment of or securing of licenses to intellectual property, product development or introduction (including, without limitation, any clinical trial accomplishments, regulatory or other filings, approvals or milestones, discovery of novel products, maintenance of multiple products in pipeline, product launch or other product development milestones), geographic business expansion, cost targets, cost reductions or savings, customer satisfaction, operating efficiency, acquisition or retention, employee satisfaction, information technology, corporate development (including, without limitation, licenses, innovation, research or establishment of third-party collaborations), manufacturing or process development, legal compliance or risk reduction, patent application or issuance goals, or goals relating to acquisitions, divestitures or other business combinations (in
33
whole or in part), joint ventures or strategic alliances; and (xxx) other measures of performance selected by the Board.
The performance goals may be based on company-wide performance or performance of one or more business units, divisions, affiliates, or business segments, and may be either absolute or relative to the performance of one or more comparable companies or the performance of one or more relevant indices. Our Board is authorized at any time in its sole discretion, to adjust or modify the calculation of a performance goal for such performance period in order to prevent the dilution or enlargement of the rights of participants, (a) in the event of, or in anticipation of, any unusual or extraordinary corporate item, transaction, event or development; (b) in recognition of, or in anticipation of, any other unusual or nonrecurring events affecting us, or our financial statements in response to, or in anticipation of, changes in applicable laws, regulations, accounting principles, or business conditions; or (c) in view of the board of director’s assessment of our business strategy, performance of comparable organizations, economic and business conditions, and any other circumstances deemed relevant. Specifically, the Board is authorized to make adjustment in the method of calculating attainment of performance goals and objectives for a performance period as follows: (i) to exclude the dilutive effects of acquisitions or joint ventures; (ii) to assume that any business divested by us achieved performance objectives at targeted levels during the balance of a performance period following such divestiture; and (iii) to exclude the effect of any change in the outstanding shares of our common stock by reason of any stock dividend or split, stock repurchase, reorganization, recapitalization, merger, consolidation,spin-off, combination or exchange of shares or other similar corporate change, or any distributions to common stockholders other than regular cash dividends. In addition, the Board is authorized to make adjustment in the method of calculating attainment of performance goals and objectives for a performance period as follows: (i) to exclude restructuring and/or other nonrecurring charges; (ii) to exclude exchange rate effects, as applicable, fornon-U.S. dollar denominated net sales and operating earnings; to exclude the effects of changes to generally accepted accounting standards required by the Financial Accounting Standards Board; (iv) to exclude the effects of any items that are “unusual” in nature or occur “infrequently” as determined under generally accepted accounting principles; (v) to exclude the effects to any statutory adjustments to corporate tax rates; and (vi) to make other appropriate adjustments selected by the Board.
Other Stock Awards. The plan administrator may grant other awards based in whole or in part by reference to our common stock. The plan administrator will set the number of shares under the stock award and all other terms and conditions of such awards.
Changes to Capital Structure. In the event there is a specified type of change in our capital structure, such as a stock split, reverse stock split, or recapitalization, appropriate adjustments will be made to (1) the class and maximum number of shares reserved for issuance under the 2018 Plan, (2) the class and maximum number of shares by which the share reserve may increase automatically each year, (3) the class and maximum number of shares that may be issued on the exercise of ISOs and (4) the class and number of shares and exercise price, strike price, or purchase price, if applicable, of all outstanding stock awards.
Corporate Transactions. Our 2018 Plan provides that in the event of certain specified significant corporate transactions (or a change in control, as defined below), unless otherwise provided in an award agreement or other written agreement between us and the award holder, the plan administrator may take one or more of the following actions with respect to such stock awards:
arrange for the assumption, continuation, or substitution of a stock award by a successor corporation;
arrange for the assignment of any reacquisition or repurchase rights held by us to a successor corporation;
accelerate the vesting, in whole or in part, of the stock award and provide for its termination if not exercised (if applicable) at or before the effective time of the transaction;
arrange for the lapse, in whole or in part, of any reacquisition or repurchase rights held by us;
34
cancel or arrange for the cancellation of the stock award, to the extent not vested or not exercised before the effective time of the transaction, in exchange for a cash payment, if any; or
make a payment equal to the excess, if any, of (A) the value of the property the participant would have received on exercise of the award immediately before the effective time of the transaction, over (B) any exercise price payable by the participant in connection with the exercise.
The plan administrator is not obligated to treat all stock awards or portions of stock awards in the same manner and is not obligated to take the same actions with respect to all participants.
Under the 2018 Plan, a corporate transaction is generally the consummation of: (1) a sale of all or substantially all of our assets, (2) the sale or disposition of more than 50% of our outstanding securities, (3) a merger or consolidation where we do not survive the transaction, or (4) a merger or consolidation where we do survive the transaction but the shares of our common stock outstanding immediately before such transaction are converted or exchanged into other property by virtue of the transaction.
Change in Control.In the event of a change in control, the plan administrator may take any of the above-mentioned actions. Awards granted under the 2018 Plan may be subject to additional acceleration of vesting and exercisability upon or after a change in control as may be provided in the applicable stock award agreement or in any other written agreement between us or any affiliate and the participant, but in the absence of such provision, no such acceleration will automatically occur. Under the 2018 Plan, a change in control is generally (1) the acquisition by any person or company of more than 50% of the combined voting power of our then outstanding stock, (2) a merger, consolidation or similar transaction in which our stockholders immediately before the transaction do not own, directly or indirectly, more than 50% of the combined voting power of the surviving entity (or the parent of the surviving entity) in substantially the same proportions as their ownership immediately prior to such transaction, (3) a sale, lease, exclusive license or other disposition of all or substantially all of our assets other than to an entity more than 50% of the combined voting power of which is owned by our stockholders in substantially the same proportions as their ownership of our outstanding voting securities immediately prior to such transaction, (4) a complete dissolution or liquidation of the company or (5) when a majority of our Board becomes comprised of individuals who were not serving on our Board on the date of the underwriting agreement related to our initial public offering, or the incumbent board, or whose nomination, appointment, or election was not approved by a majority of the incumbent board still in office.
Plan Amendment or Termination. Our Board has the authority to amend, suspend, or terminate our 2018 Plan, provided that such action does not materially impair the existing rights of any participant without such participant’s written consent. Certain material amendments also require the approval of our stockholders. No ISOs may be granted after the tenth anniversary of the date our Board adopts our 2018 Plan. No stock awards may be granted under our 2018 Plan while it is suspended or after it is terminated.
Prior Amended and Restated 2018 Equity Incentive Plan
Our Board adopted our prior Amended and Restated 2018 Equity Incentive Plan (the “Prior Plan”), in June 2018 and our stockholders approved the Prior Plan in July 2018. All references in this proxy statement to the Prior Plan shall be deemed to refer to our Amended and Restated 2018 Equity Incentive Plan, as amended, unless the context otherwise requires. As of December 31, 2018, there were outstanding stock options covering a total of 6,075,825 shares of our common stock that were granted under our Prior Plan and there were no additional shares available for grant under the Prior Plan.
No additional awards will be granted under the Prior Plan, and all outstanding awards granted under the Prior Plan that are repurchased, forfeited, expire or are canceled will become available for grant under the 2018 Plan in accordance with its terms.
Stock Awards.Our Prior Plan provides for the grant of ISOs within the meaning of Section 422 of the Code to employees, including employees of any parent or subsidiary, and for the grant of NSOs, stock appreciation rights,
35
restricted stock awards, restricted stock unit awards and other stock awards to employees, directors and consultants, including employees and consultants of our affiliates. We have granted stock options under the Prior Plan.
Plan Administration.Our Board, or a duly authorized committee of our Board, has the authority to administer our Prior Plan and is referred to as the “plan administrator” herein. The plan administrator may also delegate to one or more of our officers the authority to (1) designate employees (other than officers) to receive specified stock awards and (2) determine the number of shares subject to such stock awards. Under our Prior Plan, the plan administrator has the authority to determine award recipients, dates of grant, the numbers and types of stock awards to be granted, the applicable fair market value and the provisions of each stock award, including the period of their exercisability and the vesting schedule applicable to a stock award.
Under the Prior Plan, the plan administrator also generally has the authority to effect, with the consent of any adversely affected participant, (A) the reduction of the exercise, purchase, or strike price of any outstanding award or (B) any other action that is treated as a repricing under generally accepted accounting principles.
Stock Options.ISOs and NSOs were granted under stock option agreements adopted by the plan administrator. The plan administrator determined the exercise price for stock options, within the terms and conditions of the Prior Plan, provided that the exercise price of a stock option generally cannot be less than 100% of the fair market value of our common stock on the date of grant. Options granted under the Prior Plan vest at the rate specified in the stock option agreement as determined by the plan administrator.
The plan administrator determined the term of stock options granted under the Prior Plan, up to a maximum of 10 years. If an optionholder’s service relationship with us or any of our affiliates ceases for any reason other than disability, death or cause, the optionholder may generally exercise any vested options for a period of three months following the cessation of service. This period may be extended in the event that exercise of the option is prohibited by applicable securities laws or our insider trading policy. If an optionholder’s service relationship with us or any of our affiliates ceases due to death, or an optionholder dies within a certain period following cessation of service, the optionholder or a beneficiary may generally exercise any vested options for a period of 18 months following the date of death. If an optionholder’s service relationship with us or any of our affiliates ceases due to disability, the optionholder may generally exercise any vested options for a period of 12 months following the cessation of service.
In the event of a termination for cause, options generally terminate upon the termination date. In no event may an option be exercised beyond the expiration of its term.
Acceptable consideration for the purchase of common stock issued upon the exercise of a stock option was determined by the plan administrator and may include (1) cash, check, bank draft or money order, (2) a broker-assisted cashless exercise, (3) the tender of shares of our common stock previously owned by the optionholder, (4) a net exercise of the option if it is an NSO, (5) a deferred payment arrangement or (6) other legal consideration approved by the plan administrator.
Unless the plan administrator provides otherwise, options generally are not transferable except by will or the laws of descent and distribution.
Tax Limitations on ISOs.The aggregate fair market value, determined at the time of grant, of our common stock with respect to ISOs that are exercisable for the first time by an optionholder during any calendar year under all of our stock plans may not exceed $100,000. Options or portions thereof that exceed such limit will generally be treated as NSOs. No ISO may be granted to any person who, at the time of the grant, owns or is deemed to own stock possessing more than 10% of our total combined voting power or that of any of our affiliates unless (1) the option exercise price is at least 110% of the fair market value of the stock subject to the option on the date of grant and (2) the term of the ISO does not exceed five years from the date of grant.
36
Changes to Capital Structure. In the event there is a specified type of change in our capital structure, such as a stock split, reverse stock split, or recapitalization, appropriate adjustments will be made to (1) the class and maximum number of shares reserved for issuance under the Prior Plan, (2) the class and maximum number of shares that may be issued on the exercise of ISOs and (3) the class and number of shares and exercise price, strike price, or purchase price, if applicable, of all outstanding stock awards.
Corporate Transactions. Our Prior Plan provides that in the event of certain specified significant corporate transactions, unless otherwise provided in an award agreement or other written agreement between us and the award holder, the plan administrator may take one or more of the following actions with respect to such stock awards:
arrange for the assumption, continuation, or substitution of a stock award by a surviving or acquiring corporation;
arrange for the assignment of any reacquisition or repurchase rights held by us to the surviving or acquiring corporation;
accelerate the vesting, in whole or in part, of the stock award and provide for its termination if not exercised (if applicable) at or before the effective time of the transaction;
arrange for the lapse, in whole or in part, of any reacquisition or repurchase rights held by us;
cancel or arrange for the cancellation of the stock award, to the extent not vested before the effective time of the transaction, in exchange for no consideration or for a cash payment, if any as the plan administrator deems appropriate; and
cancel or arrange for the cancellation of the stock award in exchange for a payment equal to the excess, if any, of (A) the value of the property the participant would have received on exercise of the award immediately before the effective time of the transaction, over (B) any exercise price payable by the participant in connection with the exercise.
The plan administrator is not obligated to treat all stock awards or portions of stock awards in the same manner and is not obligated to treat all participants in the same manner.
Under the Prior Plan, a corporate transaction is generally the consummation of: (1) a sale of all or substantially all of our assets, (2) the sale or disposition of at least 50% of our outstanding securities, (3) a merger or consolidation where we do not survive the transaction, or (4) a merger or consolidation where we do survive the transaction but the shares of our common stock outstanding immediately before such transaction are converted or exchanged into other property by virtue of the transaction.
Change in Control.A stock award under the Prior Plan may be subject to additional acceleration of vesting and exercisability upon or after a change in control as may be provided in the award agreement or other written agreement between us and the participant, but in the absence of such provision, no such acceleration will occur, except as described above.Under the Prior Plan, a change in control is a transaction that qualifies as a “deemed liquidation event” as defined in our amended and restated certificate of incorporation, but excluding (1) a capitalization adjustment, (2) a public offering of our securities, (3) a capital raising transaction, (4) a transaction exclusively for the purpose of changing our domicile or corporate form, or (5) a merger, consolidation or similar transaction in which our stockholders immediately before the transaction continue to hold, directly or indirectly, at the least a majority of our combined voting power or the combined voting power of the surviving entity (as applicable) immediately following such transaction.
Plan Amendment or Termination. Our Board has the authority to amend, suspend, or terminate the Prior Plan, provided that such action does not impair the existing rights of any participant without such participant’s written consent. Certain material amendments also require the approval of our stockholders. Unless terminated sooner, the Prior Plan will automatically terminate on June 24, 2028. No stock awards may be granted under our Prior Plan while it is suspended or after it is terminated.
37
2018 Employee Stock Purchase Plan
Our Board adopted, and our stockholders approved, the 2018 Employee Stock Purchase Plan (“ESPP”) in October 2018. The ESPP became effective on October 2, 2018, in connection with our initial public offering. The purpose of the ESPP is to secure the services of new employees, to retain the services of existing employees, and to provide incentives for such individuals to exert maximum efforts toward our success and that of our affiliates. The ESPP is intended to qualify as an “employee stock purchase plan” within the meaning of Section 423 of the Code for U.S. employees.
Share Reserve. The ESPP authorizes the issuance of shares of our common stock under purchase rights granted to our employees or to employees of any of our designated affiliates. As of December 31, 2018, 1,160,000 shares of our common stock were authorized under the ESPP for issuance pursuant to purchase rights granted to our employees or to employees of any of our designated affiliates. The number of shares of our common stock reserved for issuance will automatically increase on January 1 of each calendar year, beginning on January 1, 2019 through January 1, 2028, by the lesser of (1) 1% of the total number of shares of our common stock outstanding on the last day of the calendar month before the date of the automatic increase and (2) 2,320,000 shares; provided that before the date of any such increase, our Board may determine that such increase will be less than the amount set forth in clauses (1) and (2). As of the date hereof, no shares of our common stock have been purchased under the ESPP.
Administration. Our Board administers the ESPP and may delegate its authority to administer the ESPP to our Compensation Committee. The ESPP is implemented through a series of offerings under which eligible employees are granted purchase rights to purchase shares of our common stock on specified dates during such offerings. Under the ESPP, we may specify offerings with durations of not more than 27 months, and may specify shorter purchase periods within each offering. Each offering will have one or more purchase dates on which shares of our common stock will be purchased for employees participating in the offering. An offering under the ESPP may be terminated under certain circumstances.
Payroll Deductions. Generally, all regular employees, including executive officers, employed by us or by any of our designated affiliates, may participate in the ESPP and may contribute, normally through payroll deductions, up to 15% of their earnings (as defined in the ESPP) for the purchase of our common stock under the ESPP. Unless otherwise determined by our Board, common stock will be purchased for the accounts of employees participating in the ESPP at a price$14.92 per share that is at least the lesser of (1) 85% of the fair market value of a share of our common stock on December 31, 2021, less, in the first date of an offering or (2) 85%case of the fair market value of a share of our common stock onoptions, the date of purchase.
Limitations. Employees may have to satisfy one or moreexercise price of the following service requirements before participating in the ESPP, as determined by our Board, including: (1) being customarily employed for more than 20 hours per week, (2) being customarily employed for more than five months per calendar year or (3) continuous employment with us or one of our affiliates for a period of time (not to exceed two years). No employee may purchase shares under the ESPP at a rate in excess of $25,000 worth of our commonunvested stock based on the fair market value per share of our common stock at the beginning of an offering for each calendar year such a purchase right is outstanding. Finally, no employee will be eligible for the grant of any purchase rights under the ESPP if immediately after such rights are granted, such employee has voting power over 5% or more of our outstanding capital stock measured by vote or value under Section 424(d) of the Code.
Changes to Capital Structure. In the event that there occurs a change in our capital structure through such actions as a stock split, merger, consolidation, reorganization, recapitalization, reincorporation, stock dividend, dividend in property other than cash, large nonrecurring cash dividend, liquidating dividend, combination of shares, exchange of shares, change in corporate structure, or similar transaction, the Board will make appropriate adjustments to: (1) the class(es) and maximum number of shares reserved under the ESPP, (2) the class(es) and maximum number of shares by which the share reserve may increase automatically each year, (3) the class(es)
38
and number ofoption shares subject to and purchase price applicable to outstanding offerings and purchase rights and (4) the class(es) and number of shares that are subject to purchase limits under ongoing offerings.
Corporate Transactions. In the event of certain significant corporate transactions, any then-outstanding rights to purchase our stock under the ESPP may be assumed, continued, or substituted for by any surviving or acquiring entity (or its parent company). If the surviving or acquiring entity (or its parent company) elects not to assume, continue, or substitute for such purchase rights, then the participants’ accumulated payroll contributions will be used to purchase shares of our common stock within 10 business days before such corporate transaction, and such purchase rights will terminate immediately.
Under the ESPP, a corporate transaction is generally the consummation of: (1) a sale of all or substantially all of our assets, (2) the sale or disposition of more than 50% of our outstanding securities, (3) a merger or consolidation where we do not survive the transaction and (4) a merger or consolidation where we do survive the transaction but the shares of our common stock outstanding immediately before such transaction are converted or exchanged into other property by virtue of the transaction.
ESPP Amendment or Termination. Our Board has the authority to amend or terminate our ESPP, provided that except in certain circumstances such amendment or termination may not materially impair any outstanding purchase rights without the holder’s consent. We will obtain stockholder approval of any amendment to our ESPP as required by applicable law or listing requirements.
Director Compensation
The table below shows for the fiscal year ended December 31, 2018 certain information with respect to the compensation of allnon-employeeacceleration. directors of the Company, including our Executive Chairman. In addition to serving on our Board of Directors, our Executive Chairman provides additional and ongoing oversight of us, and ongoing advice and analysis with respect to our business, business strategy and potential opportunities in the field of allogeneic CAR T cell therapy.
2018 Director Compensation Table
| | | | | | | | | | | | | | | | | | | | |
Name | | Fees
earned ($) | | | Stock
awards
($)(1) | | | Option
awards
($)(2) | | | All other
Compensation
($)(3) | | | Total ($) | |
Arie Belldegrun, M.D., FACS | | | — | | | | 8,226,852 | | | | 1,533,105 | | | | 286,226 | | | | 10,046,183 | |
David Bonderman | | | — | | | | — | | | | — | | | | — | | | | — | |
John DeYoung | | | — | | | | — | | | | — | | | | — | | | | — | |
Franz Humer, Ph.D. | | | 32,500 | | | | — | | | | 288,488 | | | | — | | | | 320,988 | |
Joshua Kazam | | | — | | | | 3,592,474 | | | | — | | | | — | | | | 3,592,474 | |
Deborah Messemer | | | 10,000 | | | | — | | | | 2,118,900 | | | | — | | | | 2,128,900 | |
Todd Sisitsky | | | — | | | | — | | | | — | | | | — | | | | — | |
Owen Witte, M.D. | | | 20,000 | | | | — | | | | 288,488 | | | | — | | | | 308,488 | |
Robert Abraham, Ph.D. (4) | | | — | | | | — | | | | — | | | | — | | | | — | |
(1) | In connection with the issuance of the Company’s Series A and SeriesA-1 convertible preferred stock in April 2018, the Company’s founders, including Dr. Belldegrun and Mr. Kazam, agreed to modify their fully vested founders’ shares of common stock outstanding to include a forfeiture restriction that lapses based on their continued service to the Company. As such, the modified founders’ shares of common stock became compensatory upon such modification. The dollar amounts in this column represent the aggregate fair value (measured as of the modification date) of the modified shares beneficially owned by Dr. Belldegrun and Mr. Kazam at the time of the modification. The dollar amounts in this column do not include an aggregate of $23,966,036 and $5,270,698 in fair value (measured as of the modification date) of modified shares that
|
39
| have a forfeiture restriction that lapses based on Dr. Belldegrun’s and Mr. Kazam’s continued service to the Company, respectively. These shares have not been included in this column as they were not beneficially owned by Dr. Belldegrun or Mr. Kazam at the time of the modification. As of December 31, 2018, the aggregate number of unvested shares outstanding under all such modified shares beneficially owned by ournon-employee directors were: Dr. Belldegrun: 2,675,818; and Mr. Kazam: 1,028,951. |
(2) | The dollar amounts in this column represent the aggregate grant date fair value of stock option awards granted in 2018. These amounts have been computed in accordance with FASB ASC Topic 718, using the Black-Scholes option pricing model. For a discussion of valuation assumptions, see Note 12 “Stock-based Compensation” to our financial statements included in our Annual Report on Form10-K for the year ended December 31, 2018. As of December 31, 2018, the aggregate number of shares outstanding under all option awards held by ournon-employee directors were: Dr. Humer: 183,750; and Ms. Messemer: 210,000. As of December 31, 2018, the aggregate number of unvested shares outstanding issued pursuant to early exercise of stock options held by ournon-employee directors were: Dr. Belldegrun: 976,500; and Dr. Witte: 183,750.
|
(3) | Amount shown represents $163,558 in consulting fees paid to Bellco Capital LLC (“Bellco”) in 2018 and an annual performance award of $122,668 paid to Bellco in 2019 for services rendered in 2018. Bellco is owned by Dr. Belldegrun, asco-trustee of the Belldegrun Family Trust, and Dr. Rebecka Belldegrun, asco-trustee of the Belldegrun Family Trust. For more information, see description of our consulting arrangement with Bellco below under “Transactions With Related Persons—Consulting Arrangements”.
|
(4) | Dr. Abraham resigned from our Board in October 2018.
|
We have reimbursed and will continue to reimburse all of ournon-employee directors for their travel, lodging and other reasonable expenses incurred in attending meetings of our Board of Directors and committees of our Board of Directors, and will pay for the travel, lodging and other reasonable expenses incurred by our employee directors to attend meetings of our Board of Directors and, as applicable, committees of our Board of Directors.
Non-Employee Director Compensation Policy
Our Board of Directors approved the followingnon-employee director compensation policy in September 2018:
TRANSACTIONS WITH RELATED PERSONSan annual cash retainer of $40,000;
an additional annual cash retainer of $12,500, $7,500 and $5,000 for service as a member of the Audit Committee, Compensation Committee and the Nominating and Corporate Governance Committee, respectively;
an additional annual cash retainer of $25,000, $15,000 and $10,000 for service as chairman of the Audit Committee, Compensation Committee and the Nominating and Corporate Governance Committee, respectively (in lieu of the committee member retainer above);
an initial option grant to purchase 54,075 shares of our common stock, vesting in 36 equal monthly installments, and an initial restricted stock unit award that may be settled for 16,275 shares of our common stock, vesting annually over a three-year period from the date of grant; and
an annual option grant to purchase 27,300 shares of our common stock, vesting in 12 equal monthly installments, and an annual restricted stock unit award that may be settled for 7,875 shares of our common stock, vesting on theone-year anniversary of the date of grant. The annual grants will be made on the date of each of our annual meetings of stockholders.
As a result of periodic review of the policy, in March 2019, our Board of Directors and Compensation Committee determined that Messrs. Bonderman and DeYoung will have sole authority to review and update the policy for 2019. Neither Mr. Bonderman nor Mr. DeYoung accept any compensation for serving on our Board of Directors. In consultation with Compensia and in light of the increase in the value of the Company’s equity, Mr. Bonderman and Mr. DeYoung approved a reduction in the amount of equity compensation to ournon-employee directors for 2019. The revised policy is set forth below:
an annual cash retainer of $40,000;
40
an additional annual cash retainer of $12,500, $7,500 and $5,000 for service as a member of the Audit Committee, Compensation Committee and the Nominating and Corporate Governance Committee, respectively;
an additional annual cash retainer of $25,000, $15,000 and $10,000 for service as chairman of the Audit Committee, Compensation Committee and the Nominating and Corporate Governance Committee, respectively (in lieu of the committee member retainer above);
an initial option grant, vesting in 36 equal monthly installments, and/or an initial restricted stock unit award, vesting annually over a three-year period from the date of grant, having an aggregate grant date value of $850,000, with the director designating the proportionate share between the initial option grant and initial restricted stock unit award prior to or on the date of grant; and
an annual option grant, vesting in 12 equal monthly installments, and/or an annual restricted stock unit award, vesting on theone-year anniversary of the date of grant, having an aggregate grant date value of $425,000, with the director designating the proportionate share between the annual option grant and annual restricted stock unit award prior to or on the date of grant. The annual grants will be made on the date of each of our annual meetings of stockholders.
Each of the equity awards described above will vest and become exercisable subject to thenon-employee director’s continuous service with us through each applicable vesting date, provided that each option and RSU award will vest in full upon a change of control of the Company, as defined under the 2018 Plan. The stock options and RSU awards will be granted under the 2018 Plan, the terms of which are described in more detail above under “- Equity Benefit Plans – Amended and Restated 2018 Equity Incentive Plan.”
Transactions With Related Persons
Related-Person Transactions policy and Procedures
We have adopted a written related-person transactions policy that sets forth our policies and procedures regarding the identification, review, consideration and oversight of “related-person transactions.” For purposes of our policy only, a “related-person transaction” is a transaction, arrangement or relationship (or any series of similar transactions, arrangements or relationships) in which we and any “related person” are participants involving an amount that exceeds $120,000. Transactions involving compensation for services provided to us as an employee, consultant or director are not considered related-person transactions under this policy. A related person is any executive officer, director, nominee to become a director or a holder of more than 5% of our common stock, including any of their immediate family members and affiliates, including entities owned or controlled by such persons.
Under the policy, where a transaction has been identified as a related-person transaction, management must present information regarding the proposed related-person transaction to our Audit Committee (or, where review by our Audit Committee would be
inappropriate, to another independent body of our Board) for review. The presentation must include a description of, among other things, all of the parties thereto, the direct and indirect interests of the related persons, the purpose of the transaction, the material facts, the benefits of the transaction to us and whether any alternative transactions are available, an assessment of whether the terms are comparable to the terms available from unrelated third parties and management’s recommendation. To identify related-person transactions in advance, we rely on information supplied by our executive officers, directors and certain significant stockholders. In considering related-person transactions, our Audit Committee or another independent body of our Board takes into account the relevant available facts and circumstances including, but not limited to:
• the risks, costs and benefits to us;
• the impact on a director’s independence in the event the related person is a director, immediate family member of a director or an entity with which a director is affiliated;
41
• the terms of the transaction;
• the availability of other sources for comparable services or products; and
• the terms available to or from, as the case may be, unrelated third parties.
In the event a director has an interest in the proposed transaction, the director must recuse
himself or herselfthemselves from the deliberations and approval.
Certain Related-Person Transactions
The following sections summarize transactions since January 1,
20182021 to which we have been a party, in which the amount involved in the transaction exceeded $120,000 and in which any of our directors, executive officers or, to our knowledge, beneficial owners of more than 5% of our capital stock, including any of their immediate family members and affiliates, including entities owned or controlled by such persons, had or will have a direct or indirect material interest, other than equity and other compensation, termination, change in control and other arrangements, which are described under “Executive Compensation” and “Director Compensation.”
Series A andA-1 Convertible Preferred Stock Financing
In April 2018, we entered into a Series A andA-1 preferred stock purchase agreement with various investors, pursuant to which we issued and sold to participating investors an aggregate of 7,557,900
Pfizer Asset Purchase Transaction
PF Equity Holdings 2 B.V. held 22,032,040 shares of our
Series common stock based on the Schedule 13D/A
convertible preferred stock and 998,225filed on September 17, 2021 with the SEC. According to the Schedule 13D/A filing, PF Equity Holdings 2 B.V. is a wholly-owned subsidiary of Pfizer Inc. (“Pfizer”) formed for the purpose of holding certain assets owned or controlled by Pfizer or its direct or indirect subsidiaries. Based on a Form 4 filed on April 4, 2022 by PF Equity Holdings 2 B.V., Pfizer held the 22,032,040 shares
as of
our SeriesA-1 convertible preferred stock at a purchase price of $35.06 per share, and received aggregate gross proceeds of approximately $300 million. Half of this funding was received in April 2018 and the remainder was received in July and August 2018.The participants in the Series A andA-1 convertible preferred stock financing included the following executive officers and members of our Board and holders of more than 5% of our capital stock or entities affiliated with them. The following table sets forth the aggregate number of shares of convertible preferred stock issued to these related parties in the Series A andA-1 convertible preferred stock financing:
| | | | | | | | | | | | |
Participants | | Shares of
Series A
Convertible
Preferred
Stock | | | Shares of
SeriesA-1
Convertible
Preferred
Stock | | | Consideration | |
Executive Officers and Directors | | | | | | | | | | | | |
David Chang, M.D., Ph.D. (1) | | | 5,704 | | | | — | | | $ | 199,995 | |
Joshua Kazam | | | 3,565 | | | | — | | | $ | 124,997 | |
Arie Belldegrun, M.D., FACS (2) | | | 27,095 | | | | — | | | $ | 950,011 | |
Owen Witte, M.D. | | | 7,130 | | | | — | | | $ | 249,994 | |
Franz Humer, Ph.D. | | | 14,261 | | | | — | | | $ | 500,023 | |
Greater than 5% stockholders | | | | | | | | | | | | |
Pfizer Inc. | | | — | | | | 998,225 | | | $ | 34,999,998 | |
Entities affiliated with TPG Carthage Holdings, L.P. (3) | | | 4,278,107 | | | | — | | | $ | 149,999,984 | |
Gilead Sciences, Inc. | | | 1,426,036 | | | | — | | | $ | 50,000,007 | |
Entities affiliated with VVAG Special Fund LLC (4) | | | 1,426,036 | | | | — | | | $ | 50,000,007 | |
Seaview Trust | | | 57,042 | | | | — | | | $ | 2,000,020 | |
(1) | Consists of 5,704 shares of Series A convertible preferred stock held by the Chang 2006 Family Trust (the “Chang Trust”). Dr. Chang, our President and Chief Executive Officer and a member of our Board, is a trustee of the Chang Trust.
|
(2) | Consists of 27,095 shares of Series A convertible preferred stock held by the Belldegrun Family Trust (the “Belldegrun Trust”). Dr. Belldegrun, a member of our Board, is a trustee of the Belldegrun Trust.
|
42
March 31, 2022.
(3) | Consists of (i) 2,852,071 shares of Series A convertible preferred stock held by TPG Carthage Holdings, L.P. and (ii) 1,426,036 shares of Series A convertible preferred stock held by The Rise Fund Carthage, L.P.
|
(4) | Consists of (i) 1,140,829 shares of Series A convertible preferred stock held by VVAG Special Fund LLC (“VVAG”), and (ii) 285,207 shares of Series A convertible preferred stock held by Vida Ventures, LLC (“Vida”). Arie Belldegrun, M.D., FACS, the Executive Chairman of our Board, is aCo-Founder and Managing Director of VVAG, Vida and certain of their affiliated entities.
|
Pfizer Asset Purchase Transaction
In April 2018, we entered into an Asset Contribution Agreement (the “Pfizer Agreement”) with Pfizer Inc. (“Pfizer”) pursuant to which we acquired certain assets and assumed certain liabilities from Pfizer, including a Research Collaboration and License Agreement with Cellectis S.A. and an Exclusive License and Collaboration Agreement with Les Laboratoires Servier SAS and Institut de Recherches Internationales Servier SAS (collectively, “Servier”), and other intellectual property for the development and administration of CAR T cells for the treatment of cancer.
As consideration for the purchased assets, we issued Pfizer 3,187,772 shares of our Series
A-1 Preferred Stock. In addition, we are required to make milestone payments upon successful completion of regulatory and sales milestones on a
target-by-target basis for certain targets, including CD19 and BCMA, covered by the Pfizer Agreement. The aggregate potential milestone payments upon successful completion of various regulatory milestones in the United States and the European Union are $30 million or $60 million per target (depending on the target, and $840.0 million for all targets), provided that we are not obligated to pay a milestone for regulatory approval in the European Union for an anti-CD19 allogeneic CAR T cell product, to the extent Servier has commercial rights to such territory. The aggregate potential milestone payments upon reaching certain annual net sales thresholds in North America, Europe, Asia, Australia and Oceania, which we refer to as the Territory, for a certain number of targets covered by the Pfizer Agreement are $325.0 million per target. Concurrently with our entry into the Pfizer Agreement, we and Pfizer entered into a letter agreement pursuant to which Pfizer granted us, in partial consideration for our milestone and royalty payment obligations under the Pfizer Agreement, an option to expand the Territory to include some or all of the rest of the world at our election. We may exercise the option at any time during the 12 year period following closing of the asset acquisition under the Pfizer Agreement.
Pfizer is also eligible to receive, on aproduct-by-product andcountry-by-country basis, (i) royalties in the low single-digit percentage on annual net sales in the United States for products commercialized by us targeting certain targets, including CD19, covered by the Pfizer Agreement, (ii) tiered marginal royalties ranging from the low tomid-single-digit percentages on annual net sales in any country in the world for products commercialized by us targeting certain other targets covered by the Pfizer Agreement and (iii) royalties in the low single-digit percentage on annual net sales in any country in the Territory for products commercialized by us
targeting targets not covered by the Pfizer Agreement that use certain Pfizer intellectual property and for which an IND is first filed on or before April 6, 2023. The royalties in the foregoing clauses (i) and (ii) are subject to reduction for products not covered by certain patent claims or for future required licenses of third party intellectual property. Our royalty obligation with respect to a given product in a given country, which we refer to as the Pfizer Royalty Term, begins upon the first sale of such product in such country and ends on the later of (i) expiration of the last claim of a defined set of patent rights, in each case covering such product in such country or (ii) 12 years from the first sale of such product in such country.
Under the Pfizer Agreement, we are required to use commercially reasonable efforts to develop and seek regulatory approval in and for the United States and the European Union for certain products covered by the Pfizer Agreement and to commercialize each product covered by the Pfizer Agreement in the applicable royalty territory in which regulatory approval for such product has been obtained. We also agreed to offer employment to certain Pfizer employees on terms no less favorable than the terms such employees enjoyed while being employed by Pfizer. We hired 39 employees from Pfizer pursuant to the terms of the Pfizer Agreement.
43
Pfizer is required, subject to certain limitations, to indemnify us against damages arising out of any breach or inaccuracy in the representations or warranties made by Pfizer, any breach of a covenant by Pfizer or any liability not acquired by us. Likewise, we are required, subject to certain limitations, to indemnify Pfizer against damages arising out of any breach or inaccuracy of our representations and warranties, any breach of a covenant made in the agreement or the related patent andknow-how license agreement by us, including any practice of intellectual property outside of the scope of the license granted to us, or any assumed liability.
In connection with the closing of the Pfizer asset purchase transaction, we entered into a Transition Services Agreement (the “TSA”) with Pfizer in April 2018, pursuant to which Pfizer provides us with certain (i) research and development services, including services relating to testing, studies, and clinical trials, project management services, laboratory equipment and operations services, animal care services, data storage services and regulatory strategy services, and (ii) general and administrative services, including business technology services, compliance services, finance/accounting services, and procurement, manufacturing and supply chain services, with respect to the assets that we purchased from Pfizer. Under the TSA, Pfizer also provides us with certain facilities and facility management services. The services are provided by certain employees of Pfizer as independent contractors of Allogene. We believe that it is helpful for Pfizer to provide such services to us under the TSA to help facilitate the efficient operation of our business after the asset purchase.
Pfizer began providing the services in May 2018 and agreed to provide the services for a period of time ranging from one to 12 months thereafter, depending on the service, which we refer to as the Service Period, with the exception of the services relating to the facilities, which Pfizer agreed to provide for up to 18 months. The services and employees for each service may be amended from time to time by the parties. Under the TSA, total expenses were $10.1 million for
During the year ended December 31,
2018 and we estimate we will pay Pfizer an aggregate of $3.82021, the Company sold $0.1 million in
2019.The TSA provides that Pfizer will indemnify us for damages that result from Pfizer’s gross negligence, willful misconduct or material breach of the TSA and that we will indemnify Pfizer for damages that arise from the provision of the services, unless such damages result from Pfizer’s gross negligence, willful misconduct or material breach. We are also requiredexcess raw materials to indemnify Pfizer for damages that arise from our material breach of the TSA.
The term of the agreement began in April 2018 and ends on the earlierPfizer.
Investors’ Rights Agreement
Pursuant to occur of the last date that Pfizer is required to provide the services or the termination of the TSA in accordance with the agreement. Either party may terminate the agreement upon 60 days’ prior written notice in the event of the other party’s uncured material breach. Pfizer may terminate the TSA upon 10 days’ prior written notice in the event of ournon-payment, if left uncured. We may terminate our use of the facilities with 60 days’ written notice.Investor Agreements
In connection with our Series A andA-1 convertible preferred stock financing, we entered into an investors’ rights agreement, dated April 6, 2018, as amended, voting agreementbetween us and rightcertain of first refusalour stockholders,
including certain holders of more than five percent of our capital stock andco-sale agreement containing registration rights, information rights, voting rights and rights of first refusal andco-sale, among other things, entities affiliated with certain of our stockholders. In addition, in connection with our saledirectors, Dr. Chang, Mr. Kazam, Dr. Belldegrun, Dr. Witte and issuance of certain convertible promissory notes (the “2018 Notes”) in September 2018, we amended our investors’ rights agreement to provide certain registration rights to the purchasers of the 2018 Notes. The foregoing agreements terminated upon the closingDr. Humer, each directors of our initial public offering, except for the registration rights set forth in the investors’ rights agreement.Consulting Arrangements
In April 2018, we entered into an Independent Contractor Agreementcompany, as well as Pfizer, entities affiliated with David Chang, M.D., Ph.D., our President and Chief Executive Officer and member of our Board, for services consistent with the role and duties of Chief Executive Officer. In exchange for the services agreed upon under the consulting agreement, we paid
44
Dr. Chang at a rate of $8,250 per week. The agreement was terminated in June 2018. We paid Dr. Chang an aggregate of $90,750 in consulting fees in 2018.
In June 2018, we entered into a letter agreement with TPG Capital – FO LLC (“TPG FO”), an affiliate of TPG Carthage Holdings, L.P. and The Riseentities affiliated with VVAG Special Fund Carthage, L.P., beneficial owners of more than 5% of our capital stock, for consulting services. PursuantLLC, are entitled to the letter agreement, TPG FO is to provide strategic, operationscertain demand registration rights, piggyback registration rights and transition consulting services for a consulting fee not to exceed $150,000 per quarter, paid in arrears beginning in April 2018, unless a higher rate is approved by our Board or our audit committee. We paid TPG FO an aggregate of $0.3 million in consulting fees in 2018.
Form S-3 registration rights.
Consulting Arrangements
In June 2018, we entered into a consulting agreement with Two River,
Consulting LLC (“Two River”). Arie Belldegrun, M.D.,
FACS, the Executive
ChairmanChair of our Board and Joshua Kazam, a member of our Board, are each partners of Two River, and David Chang, M.D., Ph.D., our President and Chief Executive Officer, is a venture partner of Two River. Pursuant to the consulting agreement, Two River provides strategic, financial, business development and secretarial consulting services and is compensated for such
services rendered at a rate of no more than $150,000 per quarter, paid in arrears beginning in April 2018, unless a higher rate is approved by our Board or our audit committee.services. We paid Two River an aggregate of $0.6 million in consulting fees in
2018.2021. Dr. Belldegrun and Dr. Chang do not receive any salary, commission or other fees for serving as partners of Two River.
In August 2018, we entered into a consulting agreement with Bellco Capital LLC (“Bellco”). Bellco is owned by our executive chairman,Executive Chair, Arie Belldegrun, M.D., FACS, asco-trustee of the Belldegrun Family Trust, and Dr. Rebecka Belldegrun, asco-trustee of the Belldegrun Family Trust, and Dr. Belldegrun is the Manager of Bellco. Pursuant to the consulting agreement, Bellco provides certain services for us, which are performed by Dr. Belldegrun and include without limitation, providing advice and analysis with respect to our business, business strategy and potential opportunities in the field of allogeneic CAR T cell therapy and any other aspect of the CAR T cell therapy business as we may agree. In consideration for these services, we paid Bellco $26,250$38,583 per month in arrears commencing June 2018 for 2018, for an aggregate of $163,558 in consulting fees in 2018.2021. In recognition of the extraordinary performance of Bellco and in helping the Companyus meet its 2018our 2021 corporate goals, an annual performance award in an amount of $122,668$0.3 million was paid to Bellco in March 2019.2022. The Company also increased the monthly payment to Bellco for 20192022 to $33,333. We$40,217. We also reimburse Bellco for out of pocket expenses incurred in performing the services. We currently have written employment letter agreements with our executive officers. For information about our employment agreements with our named executive officers, refer to “Executive Compensation—Agreements with our Named Executive Officers.”
Sublease
AgreementsAgreement
In December 2018, we subleased approximately 1,290 square feet of office space in Los Angeles, California, from Bellco. The sublease has a three-year term, which was extended to June 30, 2025, subject to certain early termination rights. Commencing in September 2018, we pay monthly base rent of approximately $6,500, increasing 3.5% per year, subject to rent abatement from November 2018 through May 2019. The monthly base rent will total approximately $220,000 for the full term of the sublease. We
also contributed to certain tenant improvements to the space totaling approximately $75,000. The monthly base rent and related occupancy costs are pass-through costs to the master landlord or other third parties and no portion is retained by Bellco.
In February 2019, we subleased approximately 2,180 square feet of our office space in New York, New York, to ByHeart, Inc., formerly known as Second Science, Inc. (“ByHeart”). ByHeart is a development stage infant formula company. Two of our Board members, Dr. Belldegrun and Mr. Kazam, have beneficial ownership in
45
ByHeart and Mr. Kazam serves on the board of directors of ByHeart. The sublease terminates in June 2025, subject to certain early termination rights. Commencing in December 2018, ByHeart pays a monthly base rent of approximately $16,000, which will total approximately $1.3 million for the full term of the sublease. The monthly base rent and related occupancy costs are pass-through costs to the master landlord or other third parties and no portion is retained by us.
Stock Options Granted to Executive Officers and Directors
We have granted stock options to our executive officers and directors, as more fully described in “Executive Compensation –
20182021 Outstanding Equity Awards Table.”
Indemnification Agreements
We have entered into, and intend to continue to enter into, separate indemnification agreements with our directors and executive officers, in addition to the indemnification provided for in our amended and restated bylaws. These agreements, among other things, require us to indemnify our directors and executive officers for certain expenses, including attorneys’ fees, judgments, fines and settlement amounts incurred by a director or executive officer in any action or proceeding arising out of their services as one of our directors or executive officers or any other company or enterprise to which the person provides services at our request. We believe that these bylaw provisions and indemnification agreements are necessary to attract and retain qualified persons as directors and officers.
The limitation of liability and indemnification provisions in our amended and restated certificate of incorporation and amended and restated bylaws may discourage stockholders from bringing a lawsuit against directors for breach of their fiduciary duties. They may also reduce the likelihood of derivative litigation against directors and officers, even though an action, if successful, might benefit us and our stockholders. A stockholder’s investment may be harmed to the extent we pay the costs of settlement and damage awards against directors and officers pursuant to these indemnification provisions.46
Director Independence
For information regarding our director and committee member independence, please see the sections above headed “Information Regarding the Board of Directors and Corporate Governance—Independence of the Board of Directors” and “Information Regarding the Board of Directors and Corporate Governance—Information Regarding Committees of the Board of Directors,” which are incorporated into this section headed “Transactions with Related Persons” by reference.
Householding of Proxy Materials
The SEC has adopted rules that permit companies and intermediaries (e.g., brokers) to satisfy the delivery requirements for Notices of Internet Availability of Proxy Materials or other annual meeting materials with respect to two or more stockholders sharing the same address by delivering a single Notice of Internet Availability of Proxy Materials or other annual meeting materials addressed to those stockholders. This process, which is commonly referred to as “householding,” potentially means extra convenience for stockholders and cost savings for companies.
This year, a number of brokers with account holders who are Company stockholders will be “householding” the Company’s proxy materials. A single Notice of Internet Availability of Proxy Materials will be delivered to multiple stockholders sharing an address unless contrary instructions have been received from the affected stockholders. Once you have received notice from your broker that they will be “householding” communications to your address, “householding” will continue until you are notified otherwise or until you revoke your consent. If, at any time, you no longer wish to participate in “householding” and would prefer to receive a separate Notice of Internet Availability of Proxy Materials, please notify your broker or the Company. Direct your written request to Allogene Therapeutics, Inc., Secretary, 210 East Grand Avenue, South San Francisco, California
94080.94080, or call us at (650) 457-2700. Stockholders who currently receive multiple copies of the Notices of Internet Availability of Proxy Materials at their addresses and would like to request “householding” of their communications should contact their brokers.
47
The Board of Directors knows of no other matters that will be presented for consideration at the
annual meeting.Annual Meeting. If any other matters are properly brought before the meeting, it is the intention of the persons named in the accompanying proxy to vote on such matters in accordance with their best judgment.
|
By Order of the Board of Directors
|
|

|
|
David M. Tanen
|
Secretary
|
|
April 24, 2019
|
By Order of the Board of Directors
David M. Tanen
Secretary
South San Francisco, California
April 26, 2022
A copy of the Company’s Annual Report to the Securities and Exchange Commission on Form
10-K for the fiscal year ended December 31,
20182021 is available without charge upon written request to: Secretary, Allogene Therapeutics, Inc., 210 East Grand Avenue, South San Francisco, California 94080.
48
APPENDIX A
CERTIFICATE OF AMENDMENT OF THE
AMENDED AND RESTATED CERTIFICATE OF INCORPORATION OF
ALLOGENE THERAPEUTICS, INC.
ALLOGENE THERAPEUTICS, INC. 210 EAST GRAND AVENUE SOUTH SAN FRANCISCO, CA 94080 VOTE BY INTERNET www.proxyvote.com Use, a corporation organized and existing under and by virtue of the Internet to transmit your voting instructionsGeneral Corporation Law of the State of Delaware (the “Company”), does hereby certify:
FIRST: The name of the Company is Allogene Therapeutics, Inc. and for electronic deliverythe date of information. Vote by 11:59 P.M. Eastern Time on June 5, 2019. Have your proxy card in hand when you accessfiling of the web site and followCompany’s original Certificate of Incorporation with the instructions to obtain your records and to create an electronic voting instruction form. ELECTRONIC DELIVERY OF FUTURE PROXY MATERIALS If you would like to reduceSecretary of State of the costs incurred by our company in mailing proxy materials, you can consent to receiving all future proxy statements, proxy cards and annual reports electronically via e-mail or the Internet. To sign up for electronic delivery, please follow the instructions above to vote using the Internet and, when prompted, indicate that you agree to receive or access proxy materials electronically in future years. VOTE BY PHONE - 1-800-690-6903 Use any touch-tone telephone to transmit your voting instructions. Vote by 11:59 P.M. Eastern Time on June 5, 2019. Have your proxy card in hand when you call and then follow the instructions. VOTE BY MAIL Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717. TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: E71441-P25456 KEEP THIS PORTION FOR YOUR RECORDS THIS PROXY CARD IS VALID ONLY WHEN SIGNED AND DATED. DETACH AND RETURN THIS PORTION ONLY ALLOGENE THERAPEUTICS, INC.State of Delaware was November 30, 2017.
SECOND: The Board of Directors recommends you vote FOR the following: 1. To elect the following Class I Director Nominees Nominees: For Against Abstain 1a. Arie Belldegrun, M.D., FACS1b. David Bonderman c. David Chang, M.D., Ph.D.The Board of Directors recommends you vote FOR the following proposalAbstainForAgainst 2.To ratify the appointment of Ernst & Young LLP as Allogene Therapeutics, Inc.’s independent registered public accounting firm for the fiscal year ending December 31, 2019NOTE: Such other business as may properly come before the meeting or any adjournment thereof.For address changes and/or comments, please check this box and write them on the back where indicated.Please indicate if you plan to attend this meeting.YesNoPlease sign exactly as your name(s) appear(s) hereon. When signing as attorney, executor, administrator, or other fiduciary, please give full title as such. Joint owners should each sign personally. All holders must sign. If a corporation or partnership, please sign in full corporate or partnership name by authorized officer.Signature [PLEASE SIGN WITHIN BOX] Date Signature (Joint Owners) Date
Important Notice Regarding the Availability of Proxy Materials for the Annual Meeting:The Notice and Proxy Statement and Annual Report are available at www.proxyvote.com. ALLOGENE THERAPEUTICS, INC.Annual Meeting of ShareholdersJune 6, 2019 10:00 AM ETThis proxy is solicited by the Board of Directors The shareholder(s) hereby appoint(s) David Chang, M.D., Ph.D. and Eric Schmidt, Ph.D., or either of them, as proxies, each with the power to appoint his substitute, and hereby authorizes them to represent and to vote, as designated on the reverse side of this ballot, all of the shares of Common Stock of ALLOGENE THERAPEUTICS, INC. that the shareholder(s) is/are entitled to vote at theAnnual Meeting of Shareholder(s) to be held at 10:00 AM ET, on June 6, 2019, at the Company’s offices located at 689 Fifth Avenue, 14th Floor, New York, New York 10022, and any adjournment or postponement thereof. This proxy, when properly executed, will be voted in the manner directed herein. If no such direction is made, this proxy will be votedCompany, acting in accordance with the Boardprovisions of Directors’ recommendations. Address Changes/Comments: (If you noted any Address Changes/Comments above, please mark corresponding box onSections 141 and 242 of the reverse side.General Corporation Law of the State of Delaware (the “DGCL”) Continued, adopted resolutions amending its Amended and Restated Certificate of Incorporation (the “Restated Certificate”), as follows:
Paragraph A of Article IV of the Restated Certificate is hereby amended and restated to read in its entirety as follows:
“A. The Company is authorized to issue two classes of stock to be designated, respectively, “Common Stock” and “Preferred Stock.” The total number of shares which the Company is authorized to issue is 410,000,000 shares. 400,000,000 shares shall be Common Stock, each having a par value of $0.001. 10,000,000 shares shall be Preferred Stock, each having a par value of $0.001.”
THIRD: This Certificate of Amendment has been duly adopted and approved by the stockholders of the Company in accordance with Sections 211 and 242 of the DGCL.
FOURTH: This Certificate of Amendment shall become effective upon filing with the Secretary of State of the State of Delaware.
IN WITNESS WHEREOF, Allogene Therapeutics, Inc. has caused this Certificate of Amendment to be signed by its President and Chief Executive Officer on reverse side
, 2022. | | | | | | | | | | | |
| ALLOGENE THERAPEUTICS, INC. | |
| | |
| By: | | |
| | David Chang, M.D., Ph.D. | |
| | President, Chief Executive Officer | |